Astronomers agree that the universe began in a Big Bang. Just how long ago that was remains a matter of debate.
Now, using a more precise chronometer, an international research team estimates the universe to be at least 12.5 billion years old, give or take 3 billion years. Previous estimates overlap with this figure but have a larger uncertainty.
The universe must be older than its oldest stars, which formed 1 to 2 billion years after the Big Bang, explains Timothy Beers of Michigan State University in East Lansing. The more precisely astronomers can determine the age of these stars, the more closely they can zero in on the age of the universe.
Astronomers have determined the age of many stars using radioactive dating. A radioactive element’s half-life is the time it takes for half of its atoms to decay into lighter so-called daughter elements. By measuring the proportions of radioactive elements and their daughters, researchers can determine how many half-lives have passed since the formation of the radioactive elements in such objects as stars. Then, it’s simple arithmetic to calculate the stars’ ages.
The method works only for stars containing radioactive isotopes. But the most ancient stars formed from just hydrogen, helium, and lithium-nonradioactive elements produced during the Big Bang. They are called metal-poor stars.
The most massive of these stars exploded, setting off nuclear reactions that fused the light elements into heavier ones that were taken up in a second generation of metal-poor stars. A small percentage of these ended up containing radioactive isotopes, says Beers.
Until now, astronomers have only been able to detect one such isotope-thorium-232-in any metal-poor star. Unfortunately, thorium-232’s half-life of 14.1 billion years is too long for precisely dating the comparably aged universe. Using this isotope to measure cosmic age is akin to timing a short race with a stopwatch marked in minutes, says Beers.
In the Feb. 8 Nature, Beers and his colleagues report finding a second radioactive isotope, uranium-238, in a metal-poor star located on the outskirts of the Milky Way. Because uranium-238’s half-life is only 4.5 billion years, it’s like having a stopwatch that ticks off seconds instead of minutes, Beers says.
Although radioactive isotopes with even shorter half-lives would allow more precise cosmic dating, they would have dwindled to nothing long ago. “Uranium is pretty close to being optimal,” says Brian Chaboyer of Dartmouth College in Hanover, New Hampshire.
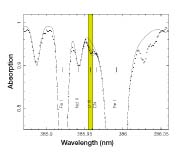
Using the Very Large Telescope in Cerro Paranal, Chile, Beers and a dozen coworkers detected spectroscopic signs of uranium in the star CS 31082-001. Different elements absorb light at different wavelengths, producing characteristic patterns in a star’s absorption spectra. The researchers then calculated the age of the star and extrapolated a minimum age of the universe.
The discovery of uranium in this star is “an important advance,” says Chaboyer. When astronomers find more of these stars, they’ll further nail down the universe’s minimum age, he says.