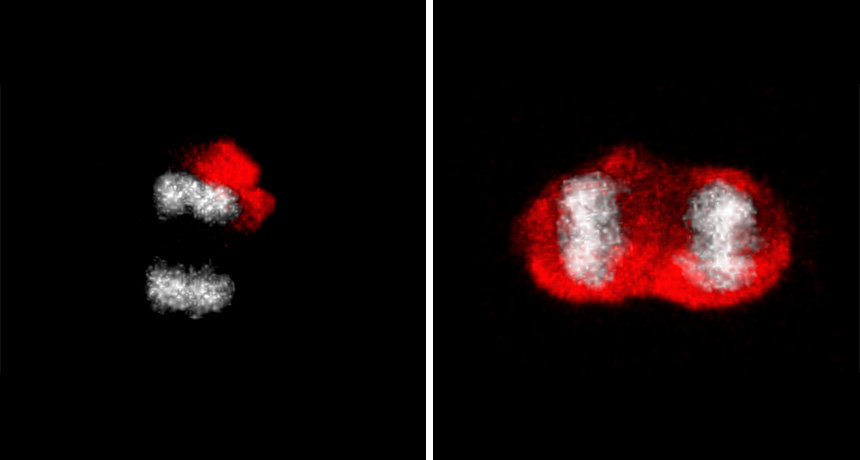
THE GREAT DIVIDE Young brain stem cells (one shown dividing, left) pack old proteins tagged with ubiquitin (red) into one daughter cell. Old cells (one shown dividing, right) lose a barrier that segregates the damaged proteins. DNA is shown in gray.
D. Moore et al/Science 2015