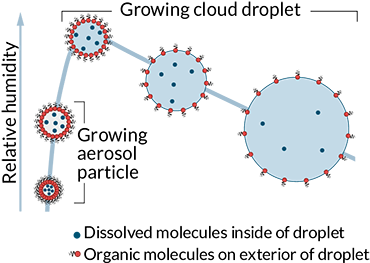
IN THE CLOUDS Many of the water droplets that make up clouds get a size boost from carbon-containing molecules that assemble on the droplets’ exterior, new research suggests.
Jan Kraus/Flickr (CC BY 2.0)
For many cloud-forming water droplets, it’s what’s on the outside that matters.
By making their own clouds, scientists have discovered a new way that fat water droplets take shape. Carbon-containing molecules envelop the exterior of developing droplets and reduce surface tension, thereby allowing more water to condense onto the drop. This effect results in droplets that are about 50 percent wider than previously expected, the researchers report in the March 25 Science.
Heftier water droplets are more apt to form clouds, says study coauthor Kevin Wilson, a physical chemist at Lawrence Berkeley National Laboratory in California. Understanding how growing droplets assemble into colossal clouds is a big deal, he says, because climate scientists currently struggle to accurately reproduce cloud formation in their climate change simulations (SN: 3/22/14, p. 22).
“A cloud model needs to capture the underlying chemistry correctly,” Wilson says. “It appears that the current one does not.”
Water droplets that make up a cloud form around airborne particles called aerosols. These aerosols, such as soot, dust and sea spray, offer a surface onto which water vapor in the air can condense. Molecules from the particles can also mix into the droplets and help them grow. For instance, water-soluble molecules such as salts from sea spray can alter the chemistry of a budding water droplet, allowing the drop to grow larger than it would with pure water alone.
Scientists assumed that this process was what controlled the size of water droplets that form around certain aerosols that contain organic molecules, such as those from fossil fuel burning, wildfires and trees. Wilson and colleagues tested that assumption by filling a roughly 4-foot-tall cylindrical tube with humid air and dropping organic aerosols into it. As water condensed and formed droplets around the aerosols, the researchers monitored droplet growth using lasers.
The seed particles, about 150 to 200 nanometers across, yielded droplets about 2,000 nanometers in diameter, the researchers found. That’s about 40 to 60 percent larger than expected had the organic molecules dissolved into the watery droplets.
After considering the chemistry of organic molecules, the researchers proposed that a previously overlooked mechanism is at play. The molecules assemble on the exterior of the water droplets, rather than mixing into the interior, the researchers suggest. Once on the surface, the molecules alter the interface between the droplets and the surrounding air, lowering surface tension and making it easier for water to condense onto the exterior of the droplet. Droplets need to reach a certain size to remain intact in the conditions typically found where clouds form, so bigger is better, Wilson says.
The work demonstrates that surface tension can’t be ignored, says Leo Donner, an atmospheric scientist at the National Oceanic and Atmospheric Administration’s Geophysical Fluid Dynamics Laboratory in Princeton, N.J. “This is important going forward for understanding the role of clouds in climate and how clouds can change as the composition of the atmosphere changes, which is another important part of climate change,” he says. Linking cloud formation to certain aerosols could help scientists identify where and how clouds will form, he says.
Moving from the small-scale chemistry of water droplets to improving global climate simulations will take time and more research, Donner says. “This isn’t the only piece of the problem, but it’s an important piece.”