Pain promoter also acts as pain reliever
Experiments with rodent cells reveal dual role for pain-sensing protein
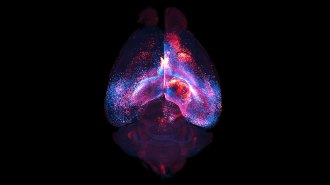
PAIN GAIN A protein that detects pain also triggers production of pain-soothing opioids, new experiments with rodent cells reveal.
ThamKC/iStockphoto
A protein that sounds the alarm when the body encounters something painful also helps put out the fire.
Called Nav1.7, the protein sits on pain-sensing nerves and has long been known for sending a red alert to the brain when the body has a brush with pain. Now, experiments in rodent cells reveal another role for Nav1.7: Its activity triggers the production of pain-relieving molecules. The study, published online January 10 in Science Signaling, suggests a new approach to pain management that takes advantage of this protein’s dual role.
“This is very interesting research,” says neuroscientist Munmun Chattopadhyay of Texas Tech University Health Sciences Center El Paso. The findings suggest that when opiates are given for certain kinds of pain relief, also targeting Nav1.7 might lessen the need for those pain relievers, Chattopadhyay says. That could reduce opiate use and their associated side effects.
The new research also solves a puzzle that has frustrated researchers and pharmaceutical companies alike. People with rare mutations in the gene for making Nav1.7 feel no pain at all. That discovery, made more than a decade ago, suggested that Nav1.7 was an ideal target for controlling pain. If a drug could block Nav1.7 activity, some kinds of pain might be eradicated (SN: 6/30/12, p 22). Yet drugs designed to do just that didn’t wipe out people’s pain.
“It seemed so obvious and simple,” says study leader Tim Hucho, a neuroscientist at the University Hospital Cologne in Germany. “But it was not so simple.”
Then in 2015, researchers reported that mice and people with nonfunctioning Nav1.7 not only felt no pain, but they also made higher than normal levels of pain-relieving opioids naturally produced by the body. When these researchers, led by John Wood of University College London, gave the opiate-blocker naloxone to a woman with the rare pain-eradicating mutation, she felt pain for the first time.
“It was astonishing,” says Hucho, whose collaborators on the new research include Wood.
Pain-sensing proteins like Nav1.7 work by prompting nerve cells to send electrical signals. But in this case, Nav1.7 was influencing a nonelectrical process — it was somehow cranking up the activity of genes in charge of making in-house opioids. “It turned the whole field upside down,” Hucho says.
An investigation of rat and mice nerve cells reveals the tug-of-war between Nav1.7’s pain-promoting and pain-relieving powers. Cells with nonfunctioning Nav1.7 have amped up activity in the cellular machinery that kicks off pain relief, Hucho and colleagues report. They suggest that Nav1.7 acts like the axis point in a playground seesaw. When the pain-promoting side is dialed down, the pain-relieving side becomes more dialed up than usual, and cells make more of their in-house opioids.
When opiates are given for pain, the body typically gets used to them and increasing amounts of the drugs are required to have an effect. Yet in the experiments with the rodent cells, this desensitization didn’t happen. The cellular machinery that interacts with the body’s homemade opioids remained sensitive to the pain relievers, even with the uptick in their production.
Taken together, the results suggest that rather than trying to push down on one side of the seesaw to stop pain, a better approach might be moving the axis at the seesaw’s center, says Hucho, tipping the scales toward in-house opioid production, while also dialing down pain promotion. The experimental design by Hucho and University Hospital Cologne colleague Jörg Isensee will make it much easier to explore how manipulations might tip the balance, the researchers say. Much more research is needed before the finding will translate into treating pain in people, but it hints at a new strategy: Rather than trying to stop pain via opiates alone, pain relief might come when such drugs are taken with a Nav1.7 blocker.
Editor’s note: This story was updated January 24, 2017, to correct the year of Johns Wood’s earlier study.