Phosphorene introduced as graphene alternative
Sheets of ultrathin phosphorus could lead to faster semiconductor electronics
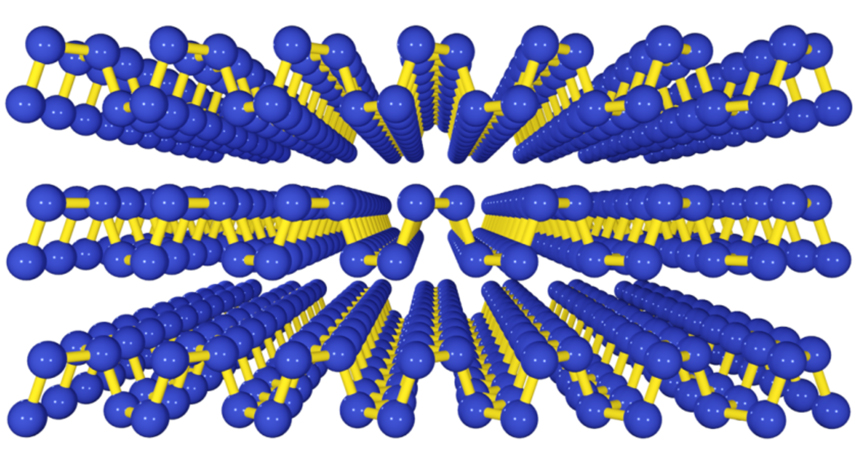
CLOSER LOOK AT PHOSPHORENE Sheets of phosphorous just one atom thick have a ridged structure, as shown in this illustration. Graphene sheets, which are made of carbon, are flat.
P. Ye