Molecules are hot. They zip, spin and vibrate with frenetic motion. They jiggle and twist on the inside and bounce on the outside, imparting structure and physical properties to nearly everything that exists. But by achieving temperatures colder than any in the natural world, physicists can almost stop these speed demons cold.

Like surgeons who slow a beating heart by packing ice around a patient’s chest, physicists have recently coaxed molecules into ultracold states in which motion is nearly gone. Researchers are left with intriguing, exquisitely controllable new specimens to poke and prod, enabling experiments that would be impossible with everyday hot molecules that rotate and vibrate at their usual frenzied pace.
To still these jittery molecules, temperatures must descend to about 350 nanokelvins, only a sliver above absolute zero and far colder even than the depths of outer space (about 3 kelvins, roughly 300 degrees Celsius colder than room temperature).
Researchers have now figured out ways to reach such lows, using precise laser pulses to trick molecules into giving up energy in the form of light. As the temperature drops, energy is siphoned away from the molecules. These new experiments have created a large, stable supply of ultracold molecules stationary enough to operate on.
“This is the breakthrough,” says Matthias Weidemüller, a physicist who was formerly at the University of Freiburg in Germany and whose group recently succeeded in making an ultracold lithium-cesium molecule. “The thing that drives the whole field is to create ultracold systems that you can manipulate and observe.”
What motivates the scientists is the potential of ultracold molecules as new tools for research. Such tools could help answer questions about the relationship between individual molecules as they collide: Cold chemical reactions are so slow that physicists have enough time to catch nuances of the interactions between molecules. Researchers could harness lattices of trapped, frozen molecules to explore the application of quantum mechanics to data storage and transmission. And more fundamentally, ultracold molecules may enable physicists to discover new, exotic phases of matter, such as a kind of superfluid in which molecules act across long ranges to influence one another in a frictionless system.
A new abundant supply of ultracold molecules has bumped physicists into their own excited state of high energy. “There is an awful lot of detail and rich physics to explore,” says Paul Julienne, a theoretical physicist at the National Institute of Standards and Technology in Gaithersburg, Md.
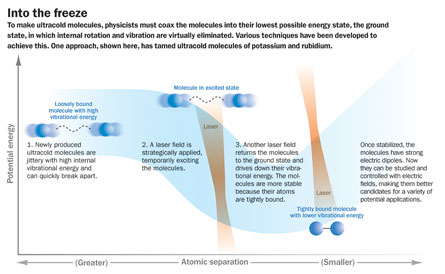
The jittery molecule
Molecules, unlike the spherical atoms that compose them, are lumpy. Physicist Jun Ye says atoms are basketballs, and molecules American footballs. Two basketballs bounce off each other in a predictable way, says Ye, of the University of Colorado at Boulder. But molecules have awkward angles and unwieldy curves, making interactions less predictable.
“Atoms are easier to control,” says Ye. “Molecules are more complex, and more exciting to study than atoms.”
Physicists have been able to freeze atoms using laser light for years. But molecules—which come in a wide variety of shapes, sizes and charges—have proved to be a greater challenge. “The techniques to ultracool atoms can’t be used for molecules,” says Weidemüller, who is now at HeidelbergUniversity. “That’s why one had to come up with a good trick to make them slow.”
Physicists had identified two approaches for creating ultracold molecules: Hot molecules could be cooled, or already cold atoms could be cajoled into joining.
Attempts by several groups started with hot molecules, but that approach proved extremely difficult. While several experiments resulted in a few weakly bound cold molecules, the molecules weren’t cold enough. They were cold on the outside but hot on the inside; they were “vibrating like hell,” says Weidemüller.
The second way—using lasers to coax already cold atoms to link up into molecules—worked better. “No one’s been able to laser-cool a molecule, but recently, people have been able to stitch together two cold atoms,” explains Julienne, who, along with colleagues including Ye and Deborah Jin, also of UC Boulder, created ultracold potassium-rubidium molecules.
Some labs, including Ye’s in Colorado and Weidemüller’s in Germany, used a magnetic field to first coax the ultracold atoms into loosely bound molecules. These proved to be the crucial starting material for ultimately taming the internal molecular motion. But, because the atoms were so far apart, the molecules weren’t stable. At the slightest provocation, they would fall apart into individual atoms. Physicists needed a way to make these “fluffy” ultracold molecules denser. The cold atoms needed to hold hands, not shout hello to each other from across the football field.
Minding the energy gap
During the transition from weakly bound—when the two atoms are loosely linked and still vibrating internally with energy—to a tightly bound molecule in its ground state, a lot of energy is released. The challenge is to remove that energy without generating any heat. Bonds between the atoms in these loose molecules are already as far apart as possible, so even a small amount of heat could push the atoms over the edge, sending them whizzing away from each other.
Ye says dropping the energy level from that of the loosely bound molecules to tightly bound ones without heating up the system is analogous to hiking from the top of the Grand Canyon down to the Colorado River without producing a drop of sweat. “This is a huge energy space to traverse,” Ye says. His team sprinted down to the bottom without perspiring, and published the results in the Oct. 10 Science.
Around the same time, other research teams also successfully created different types of stable ultracold molecules. Weidemüller’s group created molecules made of an atom of lithium and one of cesium, similar to Ye’s diatomic molecule of potassium and rubidium. Other groups created molecules composed of two atoms of cesium and molecules of two atoms of rubidium. And yet another group managed to make an ultracold molecular plasma—a form of matter with free-flowing electrons—from nitric oxide molecules. While the exact methods differed, the teams’ approaches are complementary, Weidemüller says, and largely based on laser technology.
For Ye’s energetically traveling molecules, all of the extra energy was taken away by photons—single packets of light—delivered by lasers. After painstaking experiments and calculations to figure out how much energy these ultracold molecules held, physicists knew exactly how much energy needed to be sapped and what wavelength of laser light would do the job. The first laser hit the loose molecules with a photon of light at exactly the right wavelength to excite the molecule into an intermediate high-energy state. Then the second laser pulse, of a different wavelength, caused energy to leave the molecule, dropping the molecule down into the lowest possible energy state. At this point, the two atoms locked together in a tightly linked molecule.
Physicists had previously been unable to get enough molecules into this stable and still ground state. Ye’s group managed to transform more than half of the loosely bound molecules into tight, stable molecules, creating a rich starting material that can be used to answer all sorts of new questions.
“It’s really a new frontier,” says Wolfgang Ketterle, a physicist at MIT who shared the physics Nobel Prize in 2001 for pioneering research on ultracold atoms.
The ultracold world
For many researchers, the ultracold frontier will be dominated by molecules made of two kinds of atoms, such as the lithium-cesium and potassium-rubidium molecules.
Lithium-cesium and potassium-rubidium are both polar molecules, with a positive electrical charge at one end and a negative charge at the other. These charge separations, or dipole moments, play some critical roles in the real world. For example, they are responsible for ice’s ability to float atop water. Because the water molecule is positive near the hydrogen atoms and negative near the oxygen atom, at low temperatures the molecules assemble themselves into structures that are less dense than liquid water. “The fact that certain molecules have dipole moments means that they can interact with each other in interesting ways,” says physicist Phillip Gould of the University of Connecticut in Storrs.
In the case of potassium-rubidium, the negative potassium atom interacts with the positive rubidium atom and forms a permanent separation of charge that gets stronger as the atoms are more tightly bound. These charges, which extend far beyond the edge of the molecule, are like little handles that researchers can grab with a magnetic field.
Using the positive and negative handles of these molecules to direct chemical reactions in ultraslow motion makes ultracold chemistry possible. Knowing every detail about these molecules’ charges and energy levels will allow researchers to monitor in excruciating detail exactly how two molecules interact. What’s more, because ultracold molecules can be forced to get very close to each other, the interactions between them will be different from interactions in normal chemical reactions. The subatomic nuances—the warts and hairs on the molecules that normally would be obscured by motion—will be visible. “Basically, we’re learning to control atoms and molecules in very precise ways,” Julienne says.
This control could lead to new technologies. Although Julienne calls it a “pie-in-the-sky” idea, ultracold molecules may bring physicists closer to building a quantum computer, which relies on the features of quantum physics to transfer and store information. Basing a quantum computer on ultracold, polar molecules may bring about faster, more secure computers (SN: 8/16/08, p. 24) by exploiting the long-range effects of charged molecules. “No one anticipated the speed with which these [ultracold polar] molecules would emerge. We can now seriously dream of quantum computers,” Weidemüller says. “Our kids will see quantum computing.”
Cold chemistry and quantum computing may not even be the strangest things on the ultracold molecule horizon. The strong dipole interactions that these polar molecules exhibit may allow physicists to create new forms of matter that don’t yet exist in nature, Ye says.
Weidemüller concurs: “The predictions are that there will be new kinds of phases” that arise from this breakthrough.
New, strange phases of matter may include things like a molecular supersolid, a type of matter in which the molecules would be arranged in a solid shape but could flow inside the solid without friction. The creation of a supersolid made of helium atoms was first reported in 2004, but the finding remains controversial. Also intriguing is a particular kind of matter that might form when ultracold molecules are bound to one another by their dipole interactions, says Benjamin Lev, a physicist at the University of Illinois at Urbana-Champaign. This interconnected series of molecules could have applications for superconductivity, the frictionless flow of electrons (SN: 12/20/08, p. 13). Understanding how superconductivity works at very low temperatures may ultimately lead to the creation of superconductive types of matter that would work at higher temperatures, a much-anticipated feat that would “revolutionize power grids around the world,” Lev says.
“The ultracold regime is completely new to the world, and it may produce new phases that no one even thought of,” says Lev, whose own research is aimed at exploring exotic matter formed by dipolar atoms.
With the creation of just a handful of types of stable ultracold molecules, the field of ultracold molecules has entered its infancy. “It’s one of those developments that comes along rarely but can be transformational,” says Julienne.






