Sharing the Health: Cells from unusual mice make others cancerfree
Immune-cell transplants from an extraordinary strain of mice that resists cancer can pass this trait to mice that aren’t as lucky, according to a new study.
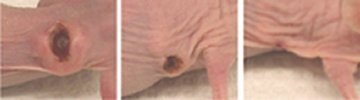
Seven years ago, Zheng Cui and Mark Willingham of Wake Forest University in Winston-Salem, N.C., and their colleagues discovered a peculiar male mouse. The researchers, who were studying tumor growth, had injected this mouse and others with lethal doses of cancer cells. While the other rodents developed fast-growing tumors, the mouse, known as number 6, remained healthy. “We thought we’d made a mistake,” says Willingham.
The mouse continued to thrive even after the scientists repeatedly injected it with a wide range of cancer-cell types in increasing amounts. Eventually, the researchers came to a surprising conclusion: The rodent was eradicating cancer cells from its body.
In breeding tests, the trait turned up in about 40 percent of the unusual mouse’s offspring.
Cui, Willingham, and their colleagues suspected that the rodents’ immune systems were fighting off disease. But they didn’t know how the animals accomplished this feat or whether the cancer resistance could be transferred to normal mice.
Since then, the scientists have examined the immune cells that swarm to cancer cells injected into the cancer-resistant animals. The majority of these responding cells are neutrophils, macrophages, and natural killer cells—the white blood cells that form the core of what’s called the innate immune system. Unlike the adaptive immune system, the innate immune system recognizes invaders on first exposure.
The researchers discovered that selectively killing off one or two of the responding cell types didn’t affect how well a cancer-resistant mouse fended off the disease. However, removing all three cell populations rendered the rodents as defenseless as normal mice.
When Cui and Willingham’s team collected white blood cells from cancer-resistant animals and transplanted them into normal mice, the recipients became cancer resistant within several days.
The effect even worked retroactively. When the researchers injected the cancer-resistant animals’ cells into normal mice that had skin tumors, the tumors vanished within weeks. The scientists noted that with one dose of cancer-resistant white blood cells, normal animals acquired cancer immunity that typically lasted for the rest of their lives.
Cui notes that the findings, reported in the May 16 Proceedings of the National Academy of Sciences, could eventually lead to drugs, cell transplants, or other therapies for cancer patients. He and his colleagues are now investigating which gene or genes are responsible for the cancer-resistant trait.
Howard Young of the National Cancer Institute in Frederick, Md., calls the report “a very intriguing paper, which raises more questions than it answers.” He notes that an understanding of the mechanism by which the mice reject tumors might provide insight into the scattered medical reports of people spontaneously recovering from cancer. However, he adds, “although success in mice is encouraging, translation into diseases in people is a long way down the road.”
Agreeing that looking for clinical applications is “superpremature,” immunologist Nora Disis of the University of Washington in Seattle says that the unusual rodents might serve as a tool for studying how the innate immune system affects cancer. “Most of the tumor-immunity world focuses on [the adaptive immune system], but there’s a burgeoning interest in the innate immune system’s role,” she adds.