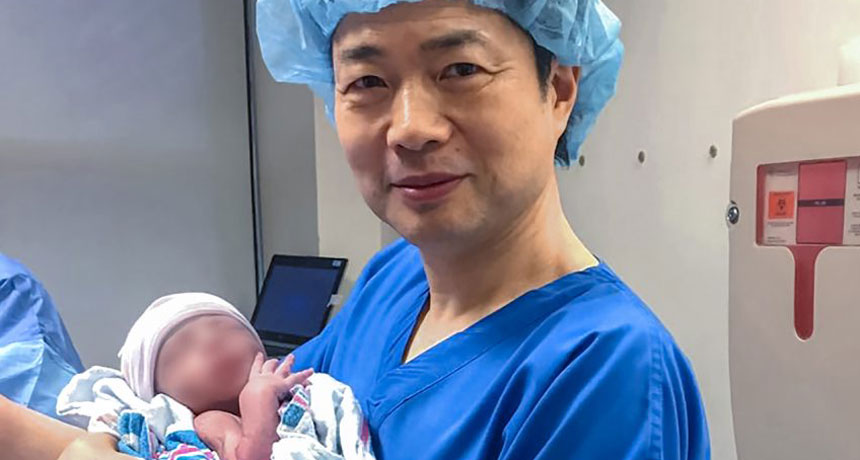
IT’S A BOY Fertility doctor John Zhang holds a baby boy (face blurred for privacy) who is the world’s first child created by a mitochondrial replacement technique called spindle transfer. Such children have been dubbed “three-parent babies.”
New Hope Fertility Center