Throat Therapy
Scientists seek a cough remedy that really delivers relief
Each year more than 26 million people in the United States go to a doctor complaining about a cough. Most have colds and will just have to wait it out. Other people cough because they have allergies, asthma, bronchitis, pneumonia or even cancer. In rare instances, there is no known culprit — a person simply starts coughing and can’t stop.


What all these patients desperately want, and what doctors don’t have, is a way to just make the cough go away. Americans end up spending $4 billion a year on over-the-counter remedies that the American College of Chest Physicians says show little evidence of effectiveness.
As common as coughing is, it turns out not to be very well understood. “Cough in general has until recently been grossly under-researched,” says pulmonologist Peter Dicpinigaitis, who heads the Montefiore Cough Center in New York City, one of the few such centers in the country. He notes that the latest new medicines approved to treat a cough, such as dextromethorphan (the active cough-suppressing ingredient in Robitussin), entered the market more than a generation ago. Over-the-counter cough suppressants are considered weakly effective, when effective at all, and prescription medicines are fraught with side effects.
But compared with previous decades, the last few years have yielded significant insights into cough science, encouraging more researchers and pharmaceutical companies to have a look. (Given how much money is spent on cough suppressants, pharmaceutical companies recognize that a better, safer syrup would be a blockbuster.)
Studies so far have found molecular on-and-off cough switches on the surface of the respiratory tract, although none have yet led to new cough medicines. New findings also show that coughing is a complicated event that shares some of the same neural circuitry with other basic body functions, like pain and temperature sensitivity. And brain scans are revealing a neural network for coughing that goes beyond a mindless impulse.
“It’s been a remarkable decade,” says Brendan Canning, a pulmonologist at the Johns Hopkins Asthma and Allergy Center in Baltimore. In 2005, a new scientific journal, called Cough, was launched in response to a sudden burst of interest in the field.
Hard to swallow
Cough medicines now on the market work in the brain, not the airways. In addition to dextromethorphan, opiates such as codeine and hydrocodone are also prescribed for cough. Both treatment types act in the brain stem by interrupting communication between nerve fibers coming from the lungs and the nerves projecting to the muscles that prompt a cough. The problem is that the target molecules that dextromethorphan and opiates attach to — their “receptors” — appear on nerve cells all over the brain, so the drugs produce other effects, such as drowsiness and labored breathing.
Current cough syrups also have the potential to be addictive, which makes policy makers and doctors hesitant to rely on them. Prescription drug abuse with opiates has become a national epidemic in the United States, and some lawmakers have moved to ban the sale of over-the-counter cough syrups to minors to prevent teens from getting high on dextromethorphan.
A new approach to cough treatment could more directly act in the airways or could still target the brain, but without such widespread side effects. But here’s the tricky part: Coughing is the body’s mechanism for defending its lungs, so a medicine can’t work too well for the body’s own good.
“There’s a devastating need for new drugs,” Dicpinigaitis says. People plagued with constant coughing get so desperate for relief, he says, that he occasionally walks into his New York office and sees suitcases in the waiting room from out-of-state patients who have Googled him and come for a visit. “Most cough is acute cough associated with the common cold,” he says. “But in a subgroup of those folks, the cold comes and goes, but the cough comes and stays.” Some of his patients have spent more than a decade watching their sleep and social lives dwindle from too much coughing.
A new cough remedy might be easier to find if coughing were an easier thing to study. Rats and mice, the standard models in medical research, don’t cough the way people do. Even animals that do readily cough go quiet when placed under anesthesia, just like people. When an awake volunteer in an MRI scanner coughs, it’s challenging to keep the person still enough to get a good image of the brain.
“It’s been hard going, and for years there was no fruit coming from people’s efforts,” says Stuart Mazzone of the University of Queensland’s St. Lucia campus in Australia. “I think for a long time it was in the ‘too hard’ basket. People just gave up.”
That is, until some accidental victories.
A hot field
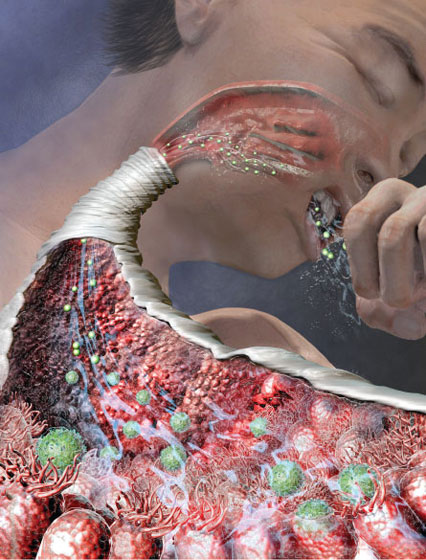
One of the early milestones in the search for new treatments came in the late 1990s, when researchers from the University of California, San Francisco announced the first discovery of a receptor that later turned out to have a role in generating a cough. Receptors are proteins that poke out from the surface of cells like icebergs, waiting for a molecule that will attach to them and set off a whole chain of events within the cell.
A substance that can trigger a cough — an irritant like smoke or a hot pepper — locks on to receptors on nerve cells after getting sucked into the airway. The joining of molecule to receptor then activates nerves that run from the respiratory system to the brain stem. Nerves in the brain stem alert nerves connecting to muscles in the chest to immediately contract. (Exactly how a cold makes you cough is still not clear, but it probably also has something to do with the irritation of nerve fibers.)
The California researchers who described the new cough receptor, called TRPV1, were not pursuing a cough molecule, but were instead interested in chili peppers. The compound capsaicin, which gives peppers their punch, is well known for its array of potent neurological effects: sweating, racing heartbeat, pain — and coughing. Research teams had been searching for the receptor that could single-handedly release such potent reactions, with the idea that it could be an attractive target for pain treatment and other problems.
“It was obvious there was going to be a receptor for capsaicin,” Canning says. And TRPV1 proved to be even more versatile than researchers had predicted. It also responds to protons, the calling card of acid rising into the esophagus. “Acid from the stomach will activate a cough,” Canning says, which is one of the reasons that people start to cough when choking.
TRPV1 generated a flurry of attention for its potential to be the target of new drugs, especially for pain. It also kicked off new interest in cough, pulling researchers into the field. Blocking the receptor, it was thought, could stop a cough at its source.
It wasn’t long before researchers developed molecules that could park themselves on the TRPV1 receptor. Soon after, Canning says, a huge glitch emerged. Besides inducing cough and sensing pain, TRPV1 has a major role in allowing the body to detect its own temperature.
Thermoregulation happens to be a hallmark of other receptors in this family of molecules. The receptor TRPM8, a distant cousin of TRPV1, was isolated in 2002. It produces a cooling sensation when activated, in the same way TRPV1 causes a burn. The yin and yang goes further. Just as TRPV1 activates cough, TRPM8 can suppress it. This explains why eucalyptus and menthol lozenges help calm some coughs — they are natural activators of TRPM8. (In fact, many public health experts believe that menthol cigarettes should be banned, because the cough-suppressing, soothing feeling of the menthol aids smoking and addiction.)
But the relief from TRPM8 activation is temporary; menthols quickly disappear from the body. What’s more, activating the receptor won’t relieve chronic cough.
TRPV1 would be a good candidate but has proved problematic. Without that receptor, the body has trouble regulating its own temperature, or detecting the temperature of outside substances. Volunteers in clinical trials were inclined to perceive the temperature of very hot liquids (such as bathwater at 120º Fahrenheit) as “optimal.” Reporting on initial tests of a TRPV1 blocker last year in the journal Pain, an international research team noted that, “it is not possible to know whether or not the observed changes in core body temperature, heat pain threshold and perception of hot liquids foretell a significant clinical risk of fever or burns.”
The success of using TRPV1 inhibitors depends on the ability to overcome the side effects of impaired thermoregulation while still blocking cough or pain, Canning says. “Quite frankly, I think a lot of companies got scared off.”
Enthusiasm for TRPV1 may be cooling, but interest in other receptors, particularly one called TRPA1, remains hot. Like those before it, TRPA1 was not discovered by researchers who had a particular interest in cough. A Swiss research team screening potential cancer drugs discovered the molecule more than a decade ago, although the scientists did not know its function or location in the body. Researchers from the Scripps Research Institute in La Jolla, Calif., and elsewhere soon found that TRPA1 was located in sensory nerves throughout the body. In 2004, a research team led by Sven-Eric Jordt, now of Yale University, reported in Nature that TRPA1 was the target of a pungent ingredient in mustard oil and wasabi. “We also found later that it was activated by a very broad range of respiratory irritants,” Jordt says, including garlic and tobacco smoke.
Research groups are now testing molecules that block TRPA1, mostly with the idea of developing pain drugs. But scientists are acutely aware that curing coughing would be a big bonus. If drugs are developed and approved for pain, Jordt says, companies would then have an easier time getting approval for cough treatment with the same compound.
A review last year in the journal Chest, from scientists at Imperial College London, called TRPA1 “one of the most promising targets currently identified for the development of novel anti-tussive drugs.” It’s still too early to know whether TRPA1 has the same drawback of interfering with the ability to detect temperature, though Jordt says animal studies so far are reassuring.
The best approach for a TRPA1 blocker as a cough suppressant may be to develop a drug that can be administered at a low dose and will target the receptors only where needed. “What I think may be better for cough is producing a formula that has to be inhaled,” Jordt says.
But targeting any particular receptor in the windpipe might still present problems of disabling a cough too much, cautions Queensland’s Mazzone. “We don’t want complete abolition of cough, but to suppress cough back to a level that is normal,” he says. Also, a healthy cough is so vital for survival that the body probably has many as yet undiscovered mechanisms to make sure it happens. One drug might have problems covering all the pathways to a cough.
Brain suppressant
Instead of looking in the airway for clues to temper a cough, Mazzone studies the brain, looking for targets beyond the brain stem. “What we are beginning to learn is that there is more to coughing than being a reflex,” he says. “A lot of the cause is behavioral. Often if you have a respiratory infection, you’ll be aware of the irritation and it will drive you nuts, but you do not cough every second. You make decisions whether to allow the cough to proceed or to voluntarily suppress it.”
When the drive to cough is not overwhelming, some mechanism in the brain is able to override the signal. This is the response that Mazzone wants to understand. “Something about the cortex and the higher brain is very important to coughing, and this hasn’t been looked at a great deal,” he says.
Using a kind of herpesvirus modified to glow fluorescent among connected nerve cells in rodents, and then examining the areas of the human brain active during a cough, Mazzone has developed a map of the brain structures involved in coughing. Writing last year in the Journal of Neuroscience, he and his colleagues described an experiment in which volunteers, their heads secured by foam padding, were given a dose of capsaicin while in an MRI scanner. It wasn’t just the brain stem that lit up during a cough, or during an urge to cough, but regions in higher parts of the brain. Other parts of the brain, such as portions of the insular cortex (an area that has a large role in depression), were more active when trying to hold back a cough.
“What we’re just starting to do now is study people with chronic cough,” says Mazzone. Volunteers with persistent coughing are being looked at with MRIs taken as they are asked to keep from coughing. They appear to show much less activity in the parts of the cortex that appear important in cough suppression. All of these results make Mazzone optimistic about one day stimulating a natural cough-suppressing mechanism.
Whatever form the next generation of cough suppressants eventually take, millions of patients are waiting, breathlessly. “The good thing about cough is that if you asked me 10 or 12 years ago what is going on, I would have said, ‘Nothing,’ ” says Dicpinigaitis. “At least now people are looking.”
Cough causes
Cough can often be relieved by treating the underlying cause, of which there are many. In some cases, though, a cough lingers for no apparent reason.
Smoking can irritate the airways and cause cough. Other irritants include air pollution, perfumes or allergens.
Asthma can cause a cough that becomes worse when a person is exposed to other irritants or cold air.
Gastroesophageal reflux disease occurs when stomach acids flow into the esophagus. These acids can trigger cough receptors.
Infections including flu, pneumonia, the common cold and whooping cough can produce a cough that sometimes lingers.
Heart failure can lead to fluid buildup in the lungs, causing chronic cough.







