Neutrons for military and medical imaging
A huge cannon shell or mortar is supposed to explode on target, not as it leaves its barrel. But if its explosive charge isn’t situated in just the right spot, the weapon could detonate at firing — wounding troops or worse. The problem: Quality control — confirming the correct positioning of a charge after a shell has been manufactured — remains challenging at best. Munitions makers would like to have the equivalent of X-rays to peer inside and confirm a charge is where it belongs. (Real X-rays won’t work, of course, because they’re blocked by the shell’s exterior metal wall.)

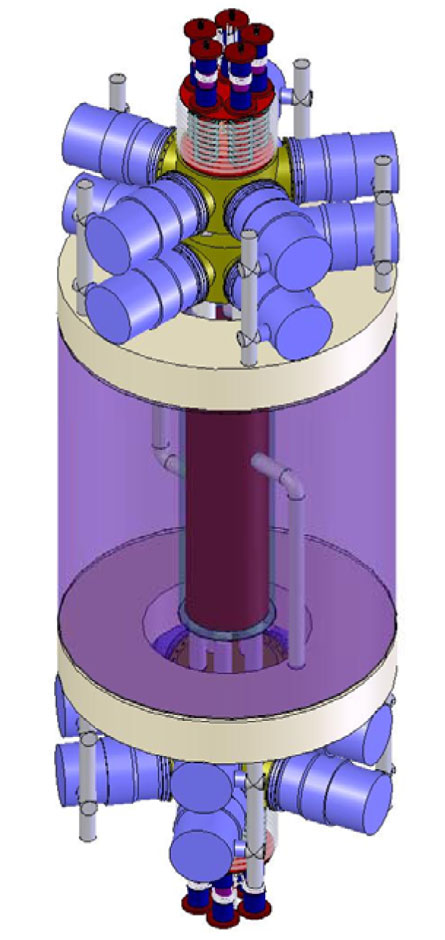
So the Army has solicited interest from the nuclear community to find a means of affordably flinging high-flux beams of neutrons at these shells. In contrast to X-rays, neutrons zip through the walls of munitions but get blocked by low-density materials inside them.
Although studies have confirmed that neutron radiography can indeed see inside big, thick-walled munitions, creating the image of a single shell’s innards can currently take hours, says Greg Piefer of Phoenix Nuclear Labs in Middleton, Wisc. The problem: The flux of neutrons in the scanning beams are more anemic than munitions makers need.
Piefer’s five-person company outside Madison, Wisc., has a $1 million Army contract to develop relatively small accelerators to speed up that imaging. Throwing 100 to 1,000 times more neutrons per second at a casing could drop the time to complete an image of that shell’s interior “into minutes or seconds,” Piefer says.
His neutron source starts with deuterium, hydrogen gas carrying a bonus neutron (present in heavy water). It’s volatilized, then bombarded with microwaves to ionize it. The deuterium nuclei then travel through a foot-long particle accelerator that speeds them up and then crashes them into gaseous tritium (a two-neutron form of hydrogen found in super-heavy water). Those deuterium-tritium collisions trigger fusion reactions that ultimately spew a slew of neutrons.
“Those neutrons then go through a layer of beryllium, which serves two purposes,” Piefer explains: It slows them down and engages them in reactions that ultimately double or triple the number of exiting neutrons.
This system does not yet exist, “But we’re building a prototype right now,” he says.
It may even offer a double payoff. The reason: A swords-to-plowshares dividend of this work could be a new technology to produce molybdenum-99, the feedstock for the most widely used nuclear isotope in diagnostic medicine. That feedstock — none of which is currently made in the United States — has been in chronically short supply for much of the past year. And it could remain so throughout the foreseeable future.
Once Piefer’s company can show that its neutron driver works reliably for at least six months at a time, he hopes to make a case to the Department of Energy and Nuclear Regulatory System that this accelerator-propelled deuterium-tritium system can safely be paired with a three-foot-diameter pool of water to make that molybdenum-99.
Dissolved within that water: a roughly 1-to-4 mix of uranium-235 to U-238. U-235 would serve as the source of molybdenum. “The U-238 is just added so you can’t make a bomb out of it,” Piefer explains. It dilutes the weapons-grade uranium (U-235) with its nonfissile cousin (U-238).
As neutrons dive into the pool, they will trigger a splitting of U-235 atoms. Some six percent of the byproduct shards will be molybdenum-99. Periodically, water would be drained from the pool and run through commercial, off-the-shelf ion-exchange resins to separate out the newly created, medical-purity molybdenum-99.
Piefer says his team has given thought to seeding the pool with molybdenum-98 and letting the neutrons fuse with these atoms to form molybdenum-99. This would allow moly makers to avoid having to work with uranium altogether. However, separating out the desired moly isotope from its medically irrelevant Mo-98 feedstock ain’t easy. Ion exchange resins don’t distinguish between them. And if the molybdenum-98 were left in the mix, Piefer says, it would increase almost 100-fold the size and weight of the already heavy medical-moly handling systems. Adding costs and headaches, that’s something to be avoided — at least now, when supplies of medical moly are tight.
So, how long should it take to have a working moly-making prototype? If all goes well, including hurdling the various regulatory obstacles to getting any new nuclear or medical-production system online, perhaps as little as four years, Piefer says. “It would take about $65 million more to get from where we are to a [moly] production facility,” he says. His company expects it could sell moly for about the same price that foreign suppliers charge today.
We’ll see. They’ve still got a lot of steps to go through to make sure that the engineering is as robust as expected and comes in on budget. But here’s an approach to domestic moly-making that may be able to piggyback its cost and much of its engineering on a program that also helps the military reliably achieve a safer bang for its buck.






