As we wait for a vaccine, here’s a snapshot of potential COVID-19 treatments

A researcher at Protein Sciences in Meriden, Conn., works on a vaccine for COVID-19, one of dozens under development.
JESSICA HILL/ASSOCIATED PRESS
Aggressive public health measures to stem the tidal wave of coronavirus infections have left people isolated, unemployed and wondering when it will all end. Life probably won’t go completely back to normal until vaccines against the virus are available, experts warn.
Researchers are working hard on that front. At least six vaccines are currently being tested in people, says Esther Krofah, chief executive of the FasterCures center at the Milken Institute in Washington, D.C. “We expect about two dozen more to enter clinical trials by this summer and early fall. That is a huge number,” Krofah said at an April 17 briefing. Dozens more are in earlier stages of testing.
In unpublished, preliminary results of a test of one vaccine, inoculated people made as many antibodies against the coronavirus as people who have recovered from COVID-19 (SN: 5/18/20). The mRNA-based vaccine induces human cells to make one of the virus’s proteins, which the immune system then builds antibodies to attack. That study was small, only eight people, but a second phase of safety testing has begun.
But vaccines take time to test thoroughly (SN: 2/21/20). Even with accelerated timelines and talk of emergency use of promising vaccines for health care workers and others at high risk of catching the virus, the general public will likely wait a year or more to be vaccinated.
In the meantime, new treatments may help save lives or lessen the severity of disease in people who become ill. Researchers around the world are experimenting with more than 130 drugs to find out if any can help COVID-19 patients, according to a tracker maintained by the Milken Institute.
Speed it up
Speed it up The Coalition for Epidemic Preparedness Innovation proposed a truncated process for COVID-19 vaccine development that replaces the lengthy, traditional linear sequence for testing, producing and licensing a vaccine (top). The new approach (bottom) involves running many steps in parallel, including ramping up manufacturing, even before knowing the vaccine will work.
Traditional vaccine development
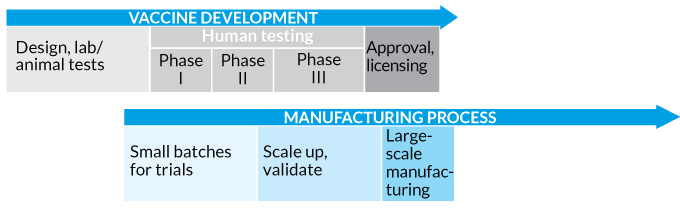
Pandemic pace
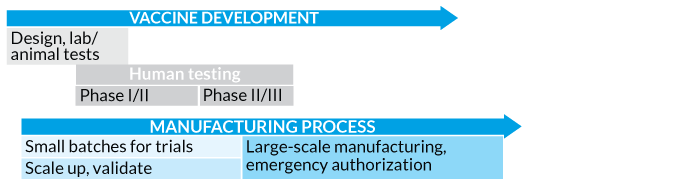
Source: N. Lurie et al/NEJM 2020
Some of those drugs are aimed at stopping the virus, while others may help calm overactive immune responses that damage lungs and other organs. Although researchers are testing a battery of repurposed drugs and devising new ones, there is still a great deal of uncertainty over whether the drugs help, or maybe even hurt.
The wait is frustrating, but there’s still much doctors and scientists don’t know about how this new coronavirus affects the body. Getting answers will take time, and finding measures to counter the virus that are both safe and effective will take even more. Early results suggest that the antiviral drug remdesivir can modestly speed recovery from COVID-19 (SN: 5/13/20). It is not a cure, but the drug may become the new standard of care as researchers continue to test other therapies.

Trustworthy journalism comes at a price.
Scientists and journalists share a core belief in questioning, observing and verifying to reach the truth. Science News reports on crucial research and discovery across science disciplines. We need your financial support to make it happen – every contribution makes a difference.
Head-on attacks
Antiviral drugs interfere with a virus’s ability to replicate itself, though such drugs are difficult to create. Remdesivir is being tested in half a dozen clinical trials worldwide. The drug mimics a building block of RNA, the genetic material of the coronavirus (SN: 3/10/20). When the virus copies its RNA, remdesivir replaces some of the building blocks, preventing new virus copies from being produced, laboratory studies have shown.
Early results in COVID-19 patients given the drug outside of a clinical trial showed that 68 percent needed less oxygen support after treatment, as reported online April 10 in the New England Journal of Medicine (SN: 4/29/20). The drug went to very sick patients, including those who needed oxygen from a ventilator or through tubes in the nose. Other researchers have disputed those results, questioning the study methods and statistical analyses, which may have given an exaggerated impression of good outcomes. The study’s authors say they have reanalyzed the data and still conclude that remdesivir has benefits.
Soon after, the U.S. National Institute of Allergy and Infectious Diseases announced that hospitalized patients with COVID-19 who got intravenous remdesivir recovered more quickly than those on a placebo: in 11 days versus 15. Those findings had not been reviewed by other scientists at the time of the announcement. The dug provides researchers with a baseline for comparing other treatments. “We think it’s really opening the door to the fact that we now have the capability of treating,” Anthony Fauci, director of the NIAID said April 29 in a news briefing at the White House.
Antiviral medications used against HIV are also being tested against COVID-19. The combination of lopinavir and ritonavir stops an HIV enzyme called the M protease from cutting viral proteins so that the virus can replicate itself. The SARS-CoV-2 virus produces a similar enzyme. But early results from a small study in China showed that the combination didn’t stop viral replication or improve symptoms (SN: 3/19/20), and there were side effects.
For now, the Society of Critical Care Medicine recommends against using the drugs, and the Infectious Diseases Society of America says patients should get the drugs only as part of a clinical trial. Several large trials may report results soon.
The HIV drugs may not work well against SARS-CoV-2, even though the viruses have similar M proteases: The coronavirus’s enzyme lacks a pocket where the drugs fit in the HIV version of the enzyme.
This illustrates why antiviral drugs are so difficult to develop. Designing a drug requires knowing the 3-D structure of the virus’s proteins, which can take months to years. But researchers are already getting some close-up views of the new coronavirus. A team in China examined the structure of the coronavirus’s M protease and designed small molecules that could block a part of the protein necessary to do its job. The team described two such molecules, dubbed 11a and 11b, April 22 in Science.
In test tubes, both molecules stopped the virus from replicating in monkey cells. In mice, 11a stuck around longer in the blood than 11b, so the researchers tested 11a further and found it seemed safe in rats and beagles. More animal tests will probably be needed to show whether it stops the virus, then multiple stages of human tests will have to follow. The drug development and testing process often takes on average 10 years or more, and can fail at any point along the way.
Meanwhile, hundreds of thousands of people worldwide have already recovered from COVID-19, and many are donating blood that might contain virus-fighting antibodies. Clinical trials are under way to test whether antibodies from recovered patients’ blood plasma can help people fight off the virus (SN: 4/25/20, p. 6). More such trials are planned.
Helping the immune system
Stopping the virus is only half the problem. In some people seriously ill with COVID-19, their immune system becomes the enemy, unleashing storms of immune chemicals called cytokines. Those cytokines trigger immune cells to join the fight against the virus, but sometimes the cells go too far, causing damaging inflammation.
Some of the drugs used to calm cytokines in cancer patients (SN: 6/27/18, p. 22) may also help people with COVID-19 ride out the storm, says cancer researcher Lee Greenberger, chief scientific officer of Leukemia and Lymphoma Society. Several of those drugs are being tested against the coronavirus now.
Hydroxychloroquine, a drug approved to treat autoimmune disorders such as lupus and rheumatoid arthritis, became a household word after President Trump touted it as a possible COVID-19 treatment.
The drug is being tested in numerous large clinical trials around the world to see if it might help calm cytokine storms in COVID-19 patients as well. But so far, there is no solid evidence that it works either to prevent infection in people or to treat people who already have the disease.
And in some studies the drug has caused serious side effects, including causing irregular heartbeats, says Raymond Woosley, a pharmacologist at the University of Arizona College of Medicine in Phoenix. People with heart problems, low potassium or low oxygen levels in their blood are at higher risk of these side effects, he says. And those are exactly the kinds of patients who are most vulnerable to COVID-19. “So, the very sickest COVID patients are those at most risk for these life-threatening arrhythmias and cardiac effects.”
Results of some rigorous clinical trials of hydroxychloroquine are expected this summer. Meanwhile, the U.S. Food and Drug Administration allows the drug to be used when no other treatment is available and patients can’t join a clinical trial.
Today’s enthusiasm for any drug that seems promising feels familiar, says Woosley. He remembers the excitement over AZT, the first drug used to fight HIV in the 1980s. It wasn’t the best drug to combat the AIDS epidemic, and better ones came later. Likewise, the first treatments for COVID-19 might be better than nothing, but not the best we will ultimately get.
Meanwhile, we wait.
With hundreds of clinical trials going on around the world, some answers may come soon. But for now, keeping the coronavirus contained will probably require aggressive testing, tracing and isolating contacts of people who have the virus and continued social distancing.
Sign up for our newsletter
We summarize the week's scientific breakthroughs every Thursday.







