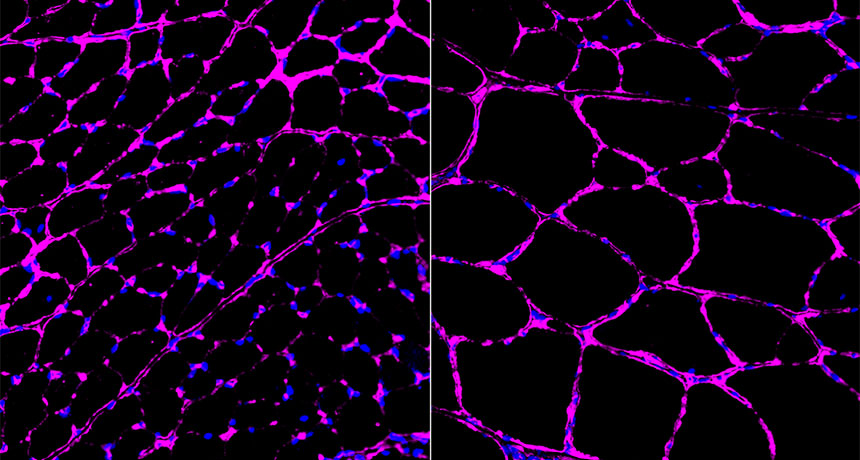
PUMP IT UP A new type of CRISPR/Cas9-based gene therapy turned on muscle-building genes in mice that have muscular dystrophy. Muscle fibers (magenta) from mice that got the therapy (right) were bigger than fibers from untreated animals (left).
H.-K. Liao et al/Cell 2017