Ebola vaccine protects people in West Africa
In Guinea trial, zero cases of virus occurred in people potentially exposed who received immediate shots
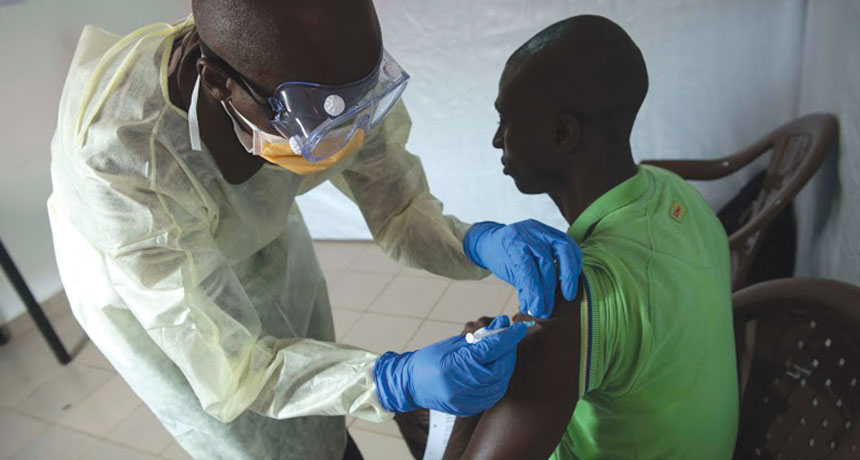
PROTECTIVE JAB A study of more than 7,000 people in Guinea shows that an Ebola vaccine guards against the lethal virus.
©Yann Libessart/MSF