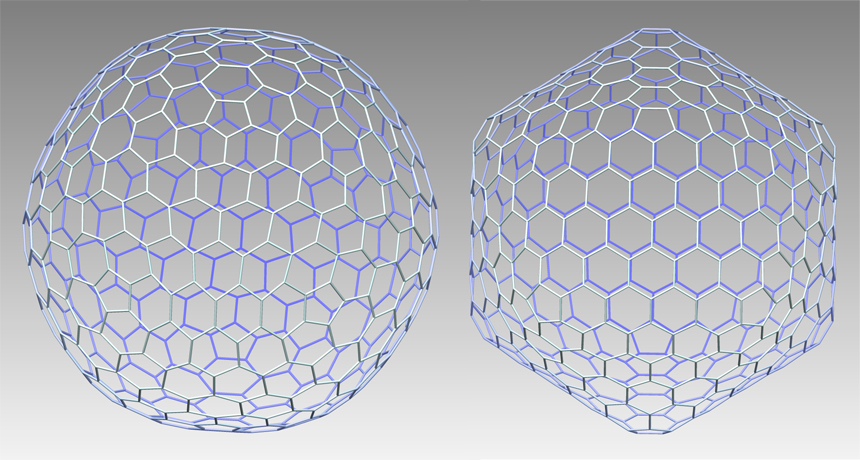
SHAPE SHIFTER A new type of molecular cage has all sides of equal length and flat faces that are all pentagons or hexagons (left). The hexagons have angles that vary from 104 to 142 degrees. A fullerene with the same number of vertices and the same pattern of faces (right) has hexagons with angles that vary in a much narrower range. This forces the hexagonal faces to warp and the cage to assume a pointy shape.
Molecular graphics performed with the University of California, San Francisco Chimera package. Figure provided by S. Schein.