The Human Brainome Project
Obama announces ambitious plan to develop new tools for exploring neural circuitry
Brain research has been on a lot of minds lately in the nation’s capital. After offering a brief shout-out to Alzheimer’s research in his February State of the Union address, President Barack Obama went a step further in April by announcing a decade-long effort to develop advanced tools for tracking human brain activity. The administration dubbed it the Brain Research through Advancing Innovative Neurotechnologies initiative, and proposed spending $100 million on the program in the 2014 fiscal year.
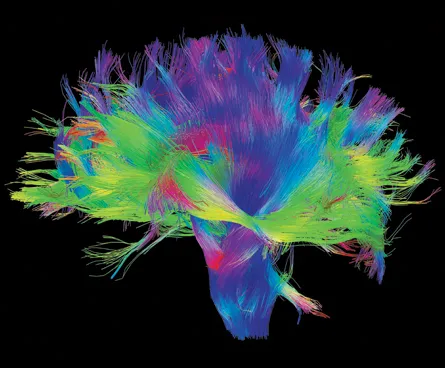
Scientists have discussed such an endeavor for years, and pushed hard for it in the past few months. Writing March 15 in Science, researchers say the project would develop technologies to probe brain activity on a far greater scale and with higher resolution than is now possible.
Current tools can monitor only small numbers of individual neurons at a time or capture blurry, bird’s-eye views of brain activity. The new tools would enable real-time mapping of how the thousands or millions of neurons in coordinated groups, known as circuits, work together. Brain functions — and, in many cases, dysfunctions — are thought to emerge from this still poorly described circuit level.
“There’s no way to build a map until you develop the tools,” says Rafael Yuste, a neuroscientist at Columbia University’s Kavli Institute for Brain Science and one of the project’s proponents.
Researchers call for developing three sets of tools to better understand brain circuits. One focus is on the creation of tools to measure the activities of all the individual neurons in a circuit. Another is on technologies to experimentally manipulate these neurons. The third tool set would store, analyze and make the data accessible to all researchers.
Scientists today can directly probe individual neurons to examine the main currency of neuronal communication, electrical signals known as action potentials. But the existing tools are generally invasive, making them tough to use in humans, or have crude resolution. New technologies, some already emerging, would be nanoscale, proponents of the effort write March 26 in ACS Nano, or they would measure voltage indirectly through an indicator. Other possible targets include chemical messengers known as neurotransmitters, which relay action potentials between neurons via synapses.
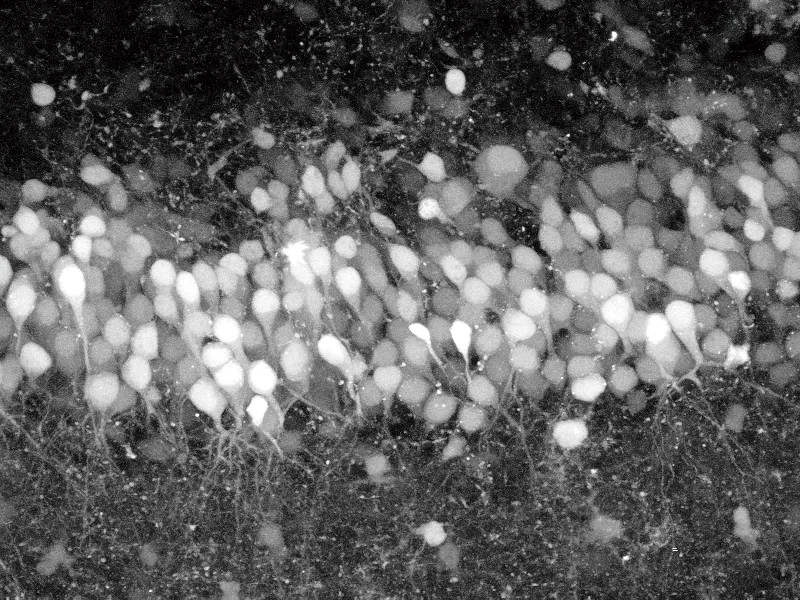
For instance, researchers already use laser microscopes to measure calcium ions, an indicator of voltage. One recent study used a special laser microscope that emits a “light sheet” to detect calcium ions and map the activity of 80 percent of a larval zebra fish’s roughly 100,000-neuron brain. Coauthor Misha Ahrens of the Howard Hughes Medical Institute in Ashburn, Va., likens the method to shining a thin sheet of light instead of a lamp in a foggy area; the thin layer would be scattered far less by the fog than the diffuse lamplight would.
The map, described March 18 in Nature Methods, shows activity once a second. It may be the first time vertebrate brain activity has ever been revealed in such detail. To go further and capture the brain’s workings at a rate of 1,000 times a second, as scientists would like, will require major changes in microscope technology, Ahrens says.
Another exciting prospect is the use of quantum dots, nanoscale semiconducting spheres that can be engineered to glow a different color or brightness depending on voltage or neurotransmitter levels.
Researchers even envision artificial cells that could serve as liaisons between measurement tools and neurons, says George Church, a Harvard University geneticist who helped plan the initiative and was a leading figure in the Human Genome Project.
Flipping switches
While imaging and measurement tools would enable researchers to link neuron activity or neurotransmitter levels with certain functions or dysfunctions of the brain, manipulating individual neurons could lead to even more powerful experiments. It also could lead to clinical applications.
In the burgeoning field of optogenetics, neurons are engineered to turn on or off in response to light. “We can selectively activate individual neurons. By doing that, you can really get at issues of causality,” says Clay Reid, a neurobiologist at the Allen Institute for Brain Science in Seattle.
Reporting April 3 in Nature, researchers at the National Institute on Drug Abuse in Baltimore and the University of California, San Francisco used optogenetics to produce or diminish compulsive cocaine use in rats by manipulating the activity of a specific group of neurons.
Researchers hope the findings lead to new therapies for drug addiction, but the road to clinical application is a difficult one and requires a sustained investment. “The evolution of optogenetics or similar techniques needs a lot of help, because the benefits are going to far, far outweigh the costs,” says coauthor Antonello Bonci of NIDA.
A huge advantage of optogenetics, he says, is that it can manipulate neurons almost in real time. But it can’t be used for long periods. In his lab, Bonci complements optogenetics with another promising technique that has lower time resolution but can be used for longer. It involves implanting neurons engineered to respond to certain compounds. Injecting those compounds can activate or silence the cells.
Preparing for the data flood
Monitoring and manipulating individual cells is only part of the challenge; tracking a million neurons a thousand times a second will produce a lot of data. Software, databases and hardware will be needed to store and distribute that information, and to process and analyze it. Project proponents met at Caltech in January to discuss how to address the data needs — roughly a gigabyte a second for a million neurons simultaneously, or 30 million gigabytes a year.
Researchers could compress the data by a factor of 10 without sacrificing crucial details, according to a report from the meeting. Ultimately, the data problem shouldn’t be insurmountable, Yuste says. Another proposed big science project, the Large Synoptic Survey Telescope, would produce around 10 million gigabytes of astronomical data annually starting in the early 2020s — right when million-neuron tools could come online, he notes.
Technical obstacles aren’t the only worry Yuste and his colleagues have. The recurring state of fiscal crisis in Washington makes it difficult to get any big project off the ground. Uncertainty over funding has fueled skepticism among scientists, who wonder whether money would be taken from other research to fund a “Big Science” project that lacks a concrete final goal.
National Institutes of Health Director Francis Collins notes that his agency has formed a workgroup of neuroscientists and some nanoscientists — supportive and skeptical alike — to guide the project’s timetable and scientific goals. One of the cochairs is Cori Bargmann, a Rockefeller University neuroscientist who previously raised concerns that the project could take funding from other neuroscience work.
Gary Marcus, a neuroscientist at New York University, says he is concerned that the project focuses too much on tool development, but notes that the administration’s proposal may be flexible enough to fund projects in other areas of neuroscience.
He fears what will happen if the tools are developed but don’t yield all the promised insights. “We will surely learn something,” Marcus says. “Whether we learn everything we want to know is another question.”







