Human cells make a soaplike substance that busts up bacteria
A surprising cellular defensive strategy could inspire new antibiotics
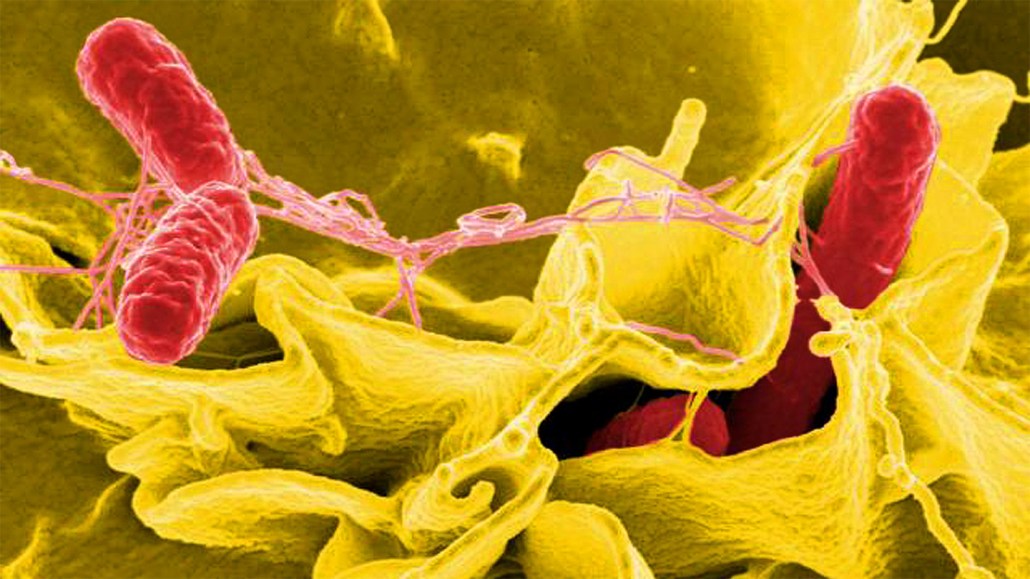
Salmonella bacteria (red in this color-enhanced micrograph) often infect the cells that line human intestines. Infected cells can fight back by releasing a detergent-like compound that helps dissolve Salmonella’s inner membrane.
NIAID