The Hunger Hormone?
An appetite stimulant produced by the stomach may lead to treatments for obesity and wasting syndromes
Perhaps you skipped breakfast this morning. It’s nearly noon and your stomach is starting to rumble. Or maybe you’re working late and developing a headache because you haven’t had dinner yet. In both of these cases, your body is sending a clear signal: Give me food, right now. Figure out how that signaling works and the world will beat a pathway to your door. Controlling weight, after all, is important for cosmetic and medical reasons, and it’s already a multibillion-dollar business.
“We all know that around mealtime, one tends to get hungry. It’s a very powerful sensation. All of us have had it. Yet the nature of that powerful stimulus is quite vague,” notes David E. Cummings of the University of Washington in Seattle. “There’s no clear consensus on why we get hungry at mealtimes.”
Over the years, scientists have proposed many hunger signals. Some researchers argued that the contracting stomach provides the trigger. One investigator went so far as to swallow a balloon and inflate it. This dulled his appetite all right, but other researchers countered that people who had had their stomachs surgically removed still became hungry.
Biologists have also debated whether a dip in the blood’s sugar concentration initiates desire for a meal. “It’s a hypothesis that has not gained universal acceptance,” Cummings says.
Now, a new candidate for a hunger signal has emerged.
Two years ago, scientists found that when they gave a hormone called ghrelin to rodents, it stimulated feeding. Last year, Cummings’ team showed that the concentration of ghrelin in a person’s blood rises significantly before meals and plummets afterwards. And in one of the latest studies, investigators observed that people receiving premeal infusions of the hormone increased their food intake by an average of 30 percent.
Although scientists only identified ghrelin in 1999, more than 100 papers on the substance have already been published. The reasons for the interest are obvious. If ghrelin is a safe appetite stimulant, the hormone could lead to treatments for wasting syndromes stemming from AIDS, cancer, heart disease, and a variety of other causes. And perhaps of greater interest to the overall U.S. population, in which obesity has become epidemic, ghrelin could become the basis for treatments that suppress appetite and provide a novel way to encourage weight loss.
“With every passing paper—and there are many per month now—the concept that ghrelin is a physiologically important regulator of appetite and body weight seems to be growing more strong,” says Cummings. “In the realm of body-weight regulation, this is one of the major stories.”
Stomach pump
Ghrelin’s discovery came about through reverse pharmacology: Synthetic drugs revealed a natural substance produced by the body.
Beginning in the late 1970s, researchers began creating ingestible drugs that stimulate the release of growth hormone, a substance known to be critical for development, tissue repair, muscle growth, bone strength, and much more.
Although growth hormone may be most vital during fetal development and childhood, it has been touted as a potential antiaging agent (SN: 5/11/96, p. 301).
While investigating some of these synthetic drugs that stimulate the release of growth hormone, researchers discovered that several don’t work through the brain’s well-known triggering agent, which is called growth hormone releasing hormone. Instead, scientists learned that the drugs act upon a previously unrecognized cell-surface protein, or receptor, in the brain’s pituitary and hypothalamus.
Biologists hypothesized that the body has a second signal, recognized by the newfound receptor, that regulates growth hormone secretion. In 1999, a research group headed by Kenji Kangawa of the National Cardiovascular Center Research Institute in Osaka, Japan, finally identified this natural signal, a peptide consisting of 28 amino acids. As expected, when injected into rodents, it stimulates growth hormone secretion.
The researchers dubbed the new molecule ghrelin. The name uses the root “ghre,” which means growth in Hindi and related languages. The name’s initial letters also reference ghrelin’s role as a growth hormone releasing factor.
Ghrelin’s discovery offered a major surprise. Although its receptors are in the brain, its primary site of production turned out to be the stomach. This suggested that the stomach pumps ghrelin into the bloodstream and the hormone then travels to the pituitary where it spurs the release of growth hormone.
Indeed, Kangawa and his colleagues found significant amounts of ghrelin in the blood of healthy people. And in 2000, Ezio Ghigo of the University of Turin in Italy and his colleagues reported that injecting people with ghrelin led to a significant, prolonged increase in growth hormone concentrations in their blood.
Anyone hungry?
Ghrelin, however, has quickly outgrown its role as a simple regulator of growth hormone secretion. Less than a year after its discovery was announced, scientists from Lilly Research Laboratories in Indianapolis, reported that injecting rodents with ghrelin prompted the animals to gain weight, most of it as fat tissue.
The hormone stimulated extra food intake by the animals and also shifted their metabolism so that they burned less fat. The investigators saw similar effects in a strain of mice unable to make growth hormone, indicating that ghrelin’s influence on weight is independent of its stimulation of growth hormone release.
Several research groups quickly confirmed the Lilly findings in rodents and began to investigate ghrelin’s role in people. Over the years, many of the people taking drugs that are now known to activate ghrelin’s brain receptors reported hunger as a side effect. And in the initial studies of ghrelin injections into people, which were designed solely to confirm that it triggers growth hormone secretion, about 80 percent of the participants complained of intense hunger.
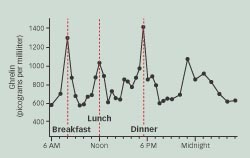
In the August 2001 Diabetes, Cummings’ group offered further evidence that ghrelin may initiate desire for food. “We reasoned that if ghrelin is a precipitant of mealtime hunger, it should rise before every meal, and as soon as you begin to eat, it should fall back down again,” says Cummings
To test that conjecture, the researchers regularly measured ghrelin in the blood of 10 healthy people over a 24-hour period. As predicted, ghrelin’s concentration rose, on average by 78 percent, before each meal and fell dramatically after the meal.
The most powerful evidence yet that ghrelin regulates human hunger comes from Stephen Bloom of Hammersmith Hospital in London and his colleagues. For one day, beginning in the morning, they continuously infused nine healthy volunteers with saline solutions either with or without ghrelin. The volunteers ate a fixed amount for breakfast 2 hours after they began their infusion and, 2 hours later, had lunch at an open buffet. Each person underwent the regimen twice, receiving ghrelin one time and not the other.
Neither the researchers nor the volunteers knew whether ghrelin had been infused until after the experiment.
Ghrelin “certainly made the human volunteers voraciously hungry,” says Bloom.
Compared with their eating when they received only saline, people infused with ghrelin ate 30 percent more, the team reports in the December 2001 Journal of Clinical Endocrinology and Metabolism.
Cummings notes, “If one Ritz cracker a day [in extra calories] would cause you to gain a pound a year, imagine what a 30 percent increase in food intake would do.”
Ghrelin is the first natural bloodborne molecule known to stimulate human appetite, concludes Bloom. Other peptides that increase appetite are made in the brain and only work if injected directly into appropriate brain regions, he explains.
Starvation signal
To their surprise, researchers have found that people suffering from wasting illnesses actually make more ghrelin than normal, not less. In fact, the highest blood concentrations of ghrelin ever recorded are in people suffering anorexia nervosa, a psychological condition in which people can literally starve themselves to death. Their bodies are apparently yelling at them to eat, but they’re not listening.
Cummings and his colleagues have also found that ghrelin concentrations in the blood follow a daily cycle, reaching high concentrations during the night. Yet hunger doesn’t prevent most people from sleeping. Cummings suspects that other biological signals change the threshold at which ghrelin triggers hunger throughout the day and night.
Last April, investigators reported in Diabetes the first results on ghrelin concentrations in the blood of obese people. The team had hypothesized that obese people overproduce ghrelin. On average, however, obese people have less ghrelin in their blood than thinner people do, the study concluded.
These results indicate that ghrelin itself isn’t the cause of most obesity. In fact, the body may naturally try to combat obesity by reducing its hunger stimulus, notes study coauthor Eric Ravussin of the Pennington Biomedical Research Center in Baton Rouge, La. An alternative explanation for obesity is that overweight people are extremely sensitive to ghrelin—for example, they may have more receptors for the hormone—so they don’t need as much of the hormone to stimulate hunger.
If labeling ghrelin as the hunger hormone is an oversimplification, how many functions does the hormone have? In just 2 years, scientists have shown that ghrelin influences blood pressure, sugar concentrations in blood, insulin metabolism, cardiac efficiency, and many other aspects of physiology. The hormone may act as a general starvation signal, preparing the body to deal with a scarcity of food, says Ghigo. To better understand ghrelin’s myriad powers, investigators are now rushing to create mice unable to make the hormone.
Cautionary tale
From a medical standpoint, the most pressing question about ghrelin is whether interfering with the hormone’s function will promote weight loss. Although most obese people already have lower-than-normal blood concentrations of ghrelin, reducing ghrelin concentration further won’t necessarily be ineffective, stresses Cummings.
There are at least three ways to inhibit ghrelin’s stimulation of appetite. One would be to suppress the stomach’s production of ghrelin. A second strategy would identify so-called ghrelin antagonists, drugs that block the hormone’s actions by binding to its receptor. While pharmaceutical companies are reportedly racing to develop such antagonists, it’s not usually easy to make drugs that can reach the brain from the bloodstream.
A third tactic could center on a unique aspect of ghrelin’s biology. For the hormone to work as an appetite stimulant, it must first attach to a small fatty acid molecule. Biologists are now searching for the enzyme that joins ghrelin to the fatty acid. Inhibiting the enzyme’s function would, in theory, leave any ghrelin made by the stomach unable to affect appetite, notes Cummings.
The most immediate medical payoff of ghrelin’s discovery may be for people suffering cachexia, a dangerous wasting induced by the loss of appetite stemming from surgery, AIDS, cancer, and other conditions. Noritoshi Nagaya of Japan’s National Cardiovascular Center says that he and his colleagues already plan to test ghrelin’s ability to stimulate appetite in patients with cachexia as a result of heart disease. This weight loss and weakening is a major contributor to the mortality of such patients, the investigator notes.
Some researchers warn against overenthusiasm about ghrelin’s prospects. Mark Friedman of Tulane University of Louisiana in New Orleans, who contends that signals from the liver regulate food intake, points to the story of leptin as a cautionary tale. Quickly dubbed the obesity hormone by the media and some scientists after its discovery, leptin is secreted by fat cells and appears to tell the brain whether the body has enough energy stored as fat (SN: 7/29/95, p. 68).
Excited by this discovery, some researchers speculated that extra leptin could trick the body into thinking that it didn’t need to eat much. A biotech company even paid $25 million for the patent rights to leptin. It’s turned out, however, that obese people make abnormally high amounts of the hormone and that providing them with more leptin doesn’t instigate significant weight loss.
Efforts to develop ghrelin into a blockbuster drug for weight management could suffer the same fate. But even if it doesn’t lead to best-selling drugs, its recent arrival on the scientific scene has whetted researchers’ appetites to learn more.







