Timing is everything. Just ask a comedian, trapeze artist, Romeo and Juliet — or nearly any cell in your body.

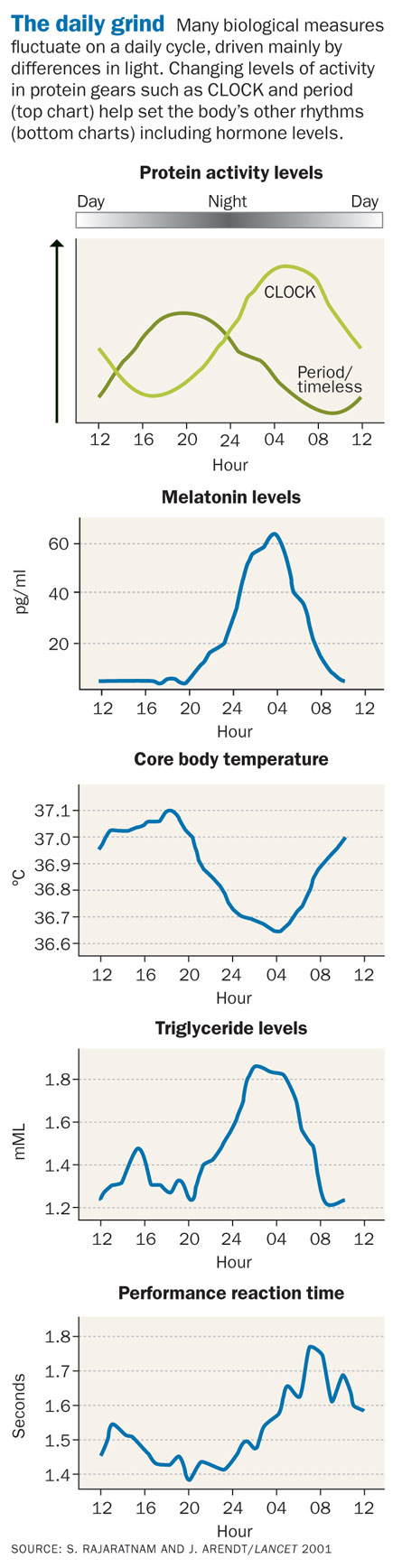
Ticking away inside almost all cells are tiny clocks composed of protein gears. Scientists have known that these molecular clocks govern the daily rhythms of life, from mealtimes and bedtimes to the rise and fall of hormone levels, body temperature and blood pressure. New research shows that circadian clocks, as the daily timekeepers are known, do more than just control day-to-day schedules.
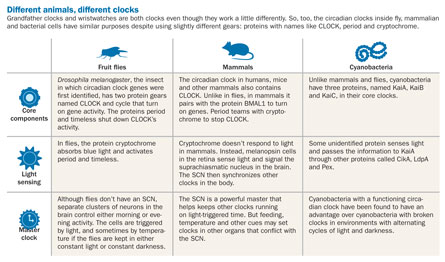
Such clocks, some scientists say, have the potential to play a role in nearly every biological function. Studies of bacteria, rodents and fruit flies suggest that circadian clocks may time processes as diverse as cellular division and aging. “When you start asking, ‘what does the clock control?’ you have to say, ‘everything,’” says Erik Herzog, a biologist at Washington University in St. Louis.
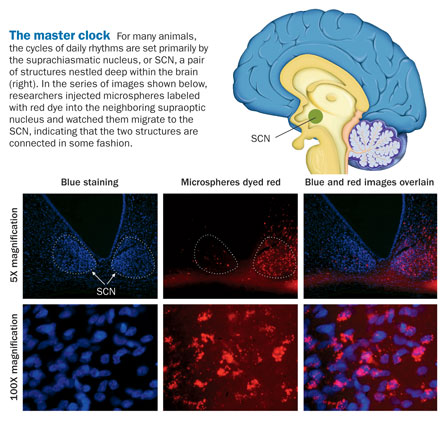
Some of the new insights come from studying the brain’s master clock, a pair of structures known as the suprachiasmatic nucleus, or SCN, that set the body’s daily rhythms. Other work, meanwhile, suggests that the SCN is not a single monolithic clock but more a set of interrelated nodes that help coordinate clocks throughout the body. And still other researchers have found that the SCN may not even be the ultimate arbiter of the body’s time, and that other organs control biological rhythms on their own without much, if any, help from the SCN.
The master’s orders
For decades, the SCN reigned supreme in scientists’ understanding of circadian clocks. A total of only 20,000 neurons populate the structures — one on each side of the brain — but that meager number of brain cells is enough to set the daily rhythms for the entire body. It’s as if the residents of a small town in New England determined the schedule of every person on Earth, and then some.
“I think of the SCN as the atomic clock in this network of ATMs,” Herzog says. Each body cell has its own clock, just as individual cash machines do. And periodically those peripheral clocks check in with a central timekeeper to stay synchronized.
The rhythm of the SCN is set mainly by light, as special cells in the retina relay a message to the brain when they sense daylight. The light cues affect the gears of the molecular clock inside the SCN’s neurons, which then somehow control the cells’ firing rates — the frequency at which neurons zip off electrical messages to other brain cells.
“We know that the clock is there [in the brain]. We’ve known about different firing rates. We’ve learned a lot about the genes that control clock activity, but none of that tells us how the clock actually controls anything,” says Charles Bourque, a neuroscientist at McGill University in Montreal.
To help tackle that question, Bourque’s team has deciphered some of the chemical messages the SCN uses to regulate a circadian rhythm that keeps the body from dehydrating during sleep.
One of the body’s main defenses against dehydration is a hormone called vasopressin, which signals the kidneys to retain water. Scientists have known for years that the hormone is released toward the end of the sleep period by neurons in the supraoptic nucleus, clusters of cells in the hypothalamus just a few millimeters from the SCN. And researchers had also worked out that when a person or animal is awake, neurons in another nearby structure known as the organum vasculosum lamina terminalis, or OVLT, sense the loss of water and signal the release of vasopressin, and induce thirst. But no one knew how the brain clock scheduled the release of vasopressin during sleep.
It turns out that SCN cells use vasopressin to help regulate the hormone’s own release during sleep, Bourque and McGill colleague Eric Trudel discovered. The researchers removed a thin slice from a rat’s hypothalamus that contained the SCN, the supraoptic nucleus and the OVLT all together. The team recorded and manipulated electrical activity in the brain regions, and found that when firing rates of clock neurons were high, communication between the OVLT and the supraoptic nucleus weakened. Conversely, as clock neurons quieted down — as they do at the end of the sleep period —communication between the other two brain regions strengthened.
The researchers concluded that some brain chemical released from the SCN was stifling communication between the other two neuron clusters for most of the time when the rat was asleep. Eliminating other candidate chemicals suggested that it must be vasopressin working with some other yet-to-be-identified substance, the team reported online February 28 in Nature Neuroscience. Vasopressin is made by about a third of SCN neurons.
In addition to vasopressin, scientists have been on the trail of mysterious substances secreted by the master clock for some time now. When researchers transplant the SCN from a rat with a working clock to another whose clock has been disabled, the new clock can set circadian rhythms, even though it isn’t physically connected to the animal’s original pacemaker. That tells scientists that the SCN must be giving off some synchronizing substance that diffuses out and regulates other brain circuits, says neuroscientist Rae Silver of Columbia University and Barnard College in New York City.
No one has tracked down that substance yet, although researchers at the University of Illinois at Urbana-Champaign may be getting close. The master clocks in rats’ brains make at least 102 different peptides — short proteins or pieces of proteins — the Illinois researchers say in the February Molecular & Cellular Proteomics. Those include 33 newly identified peptides and 12 others that are modified in different ways after being made, all of which are potential candidates for the clock-synchronizing substance.
Cooperative control
Perhaps finding the master regulator that the SCN uses to synchronize other clocks isn’t so easy because it isn’t one clock. Herzog and his colleagues have discovered that no clock neuron is an island. Alone, each clock neuron has about a 25 percent chance of maintaining a circadian rhythm for several days, the researchers reported September 22 in the Proceedings of the National Academy of Sciences. But when the cells work together, they stay reliably on beat. The result indicates that within the master clock, there is no class of master pacemakers. Instead, all cells cooperate to generate a precise daily rhythm.
Scientists have also recently discovered that the SCN is actually a clock confederation with several different clusters, or nodes. The nodes form a relay team, passing electrical activity from one to another. “One node fires, then the second, third and so on, and then the whole thing is quiet for a few minutes,” Silver says. Having independent nodes instead of one monolithic clock may make the brain clock more robust and allow it to adjust more quickly to environmental changes, Silver and her Columbia University colleague Matthew Butler argue in the October Journal of Biological Rhythms.
Still other research has found that different groups of neurons contained within the SCN behave surprisingly differently from each other. For instance, some cells within the master clock of mice (and presumably other mammals, including humans) make period 1, one of the core period proteins of molecular clocks found inside cells. Levels of the protein peak during the day and are low at night. Firing patterns of SCN neurons do the same thing, so researchers thought that levels of period 1 set the brain cells’ firing patterns.
But neurons that produce period 1 don’t behave as expected. Hugh Piggins and Mino Belle of the University of Manchester in England and colleagues measured electrical activity in master clock cells from a mouse brain, comparing cells that make period 1 with cells that don’t. The researchers found that cells without period 1 followed a firing pattern that peaked during the day and fell at night.
But cells that make the protein fired at a moderate rate in the morning, then became so overexcited in the afternoon that they could no longer fire an electrical signal, before recovering again about dusk, the team reported last October in Science (SN Online: 10/8/09). That prolonged period of overexcitation would normally allow a toxic buildup of calcium, killing most cells, but the period 1 neurons go through this near-death experience every day seemingly unscathed. The researchers don’t know why the neurons survive, nor how period 1 and the turning of other molecular gears trigger the firing pattern.
Beyond the brain
While many researchers continue to probe the mysteries of the brain clock, others think it gets too much credit for controlling circadian rhythms in the rest of the body. The liver, for instance, has a clock that works just fine without much input from the SCN, says Satchidananda Panda, a geneticist and biologist at the Salk Institute for Biological Studies in La Jolla, Calif.
Nearly 5,000 genes in the mouse liver follow a circadian cycle that corresponds to mealtimes, Panda and his colleagues reported in the Dec. 15 Proceedings of the National Academy of Sciences. Of those genes, only nine maintained the same cadence regardless of feeding or fasting, indicating that those genes are probably controlled by signals from the brain clock. Another 368 of those 5,000 genes continued to follow a circadian rhythm when mice fasted, a sign that those genes are controlled by the molecular clocks inside liver cells. But the rhythms of the vast majority of genes active in the liver are governed by eating alone, Panda says.
These results could have implications for health, he says. Many scientists think that one role for circadian clocks is to separate biological processes, such as digesting food and replicating DNA; multitasking such activities at the same time could lead to DNA damage.
Mice usually eat about 65 percent of their calories at night, when the rodents are normally active, and consume the remaining 35 percent in “midnight snacks” throughout the day. “Mice eat a little bit like modern humans,” Panda says. “They have a big dinner, take a nap, eat a little bit more.”
Panda’s team compared circadian rhythms of those mice with mice that ate the same amount but got access to food for only eight hours a day, fasting the rest of the time. Fasting led to higher peaks and lower troughs in the cyclic waves of activity levels of more than 3,000 genes, the researchers found — including genes known to encode clock proteins.
Having a more crisply defined circadian rhythm could help improve the efficiency of metabolism, Panda says. For instance, mice that can eat any time they want burn some of the sugar in their food and store the rest as fat. In contrast, mice on a restricted feeding schedule burn sugar intensely, but then switch to burning fat while fasting and rapidly return to burning sugar after eating. Without fasting, mice never really get a chance to burn fat. The genes that control those processes are among those whose rhythms follow a circadian pattern set by feeding.
“All of this boils down to that you need fasting,” Panda says. He and his colleagues are doing experiments in mice to determine whether sharpening circadian rhythms, by restricting feeding to certain times of day, will have beneficial effects on health over the long-term.
New data from studies of fruit flies also suggest that robust circadian rhythms are one key to a long and healthy life.
Many scientists assume that circadian clocks would give organisms an evolutionary advantage. “Organisms need a mechanism to predict changes in the environment before they occur,” says Michael Nitabach, a physiologist and geneticist at Yale School of Medicine. “Any organism that can predict what’s going to happen is going to have a selective advantage over an organism that can’t.”
Yet somehow reindeer seem to get along without working clocks, according to a study published March 23 in Current Biology. And fruit flies that don’t have functioning circadian clocks seem to do just as well when raised in a laboratory as flies with fully functioning clocks, says Natraj Krishnan, a physiologist at Oregon State University in Corvallis. But in the wild, flies may encounter stresses that could change the game, so Krishnan and his colleagues decided to expose lab flies to a mild stress and see if having a clock made a difference.
The researchers exposed normal flies and flies lacking the clock gene period to one day of high oxygen, which can set off harmful chemical reactions that damage DNA and proteins. Neither group of flies was bothered much by oxygen stress if the flies were exposed when young. But middle-aged flies lacking period did not fare as well. Compared with normal flies, when exposed at 35 days old the mutant flies had a 20 percent shorter life span. Oxygen-damaged proteins also built up faster in elderly mutant flies than in control flies, the researchers reported in the November Aging.
These results indicate that circadian clocks may help control more than just daily rhythms of life, Krishnan says. “I’m pretty sure that anything that messes up the phase of the clock probably affects aging and life span,” he says.
So daily clocks might be one of the important things determining when it’s “your time.” After all, circadian rhythms tend to wind down in elderly people and rodents. Further research may suggest ways to reset the health and aging clock.
And that, in turn, could underscore yet again the most important lesson of circadian clocks: that timing is everything.







