In 2019, a ketamine-based antidepressant raised hopes and concerns
The benefits and risks of Spravato are not yet clear
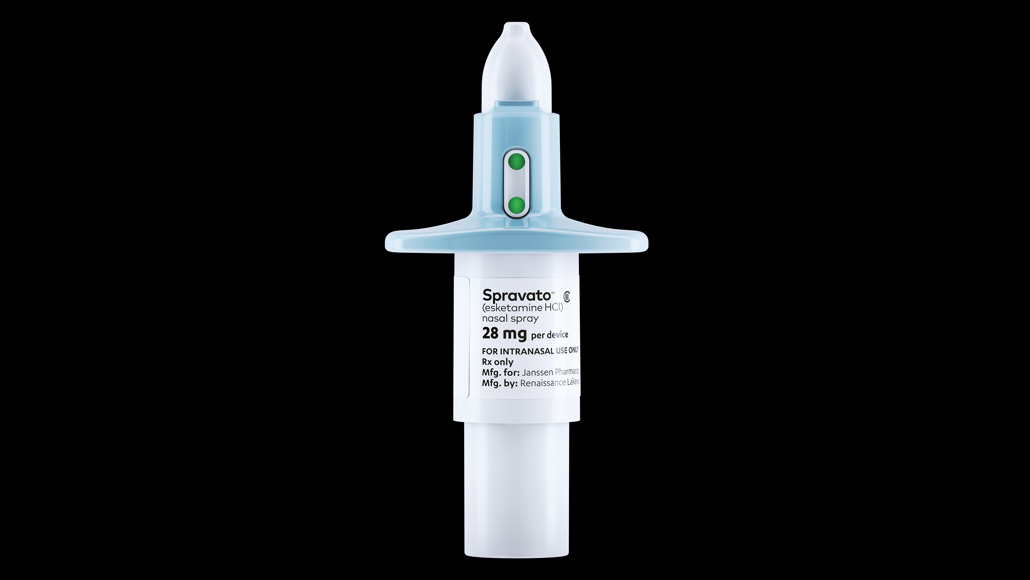
The antidepressant Spravato can be taken only at certified health clinics, to allow for close patient monitoring.
Janssen Pharmaceuticals, Inc.