Light whips platinum into shape
Researchers have created a platinum foam that can catalyze the splitting of water. John Shelnutt of Sandia National Laboratories in Albuquerque and his collaborators embedded light-absorbing molecules called porphyrins in the membranes of water-filled vesicles called liposomes. Then, the researchers placed these liposomes in an acidic solution containing platinum ions.
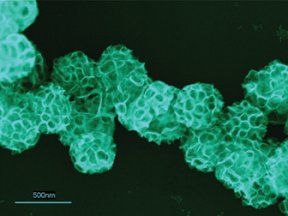
When energized with light from an overhead projector lamp, the porphyrins initiated a series of reactions resulting in the conversion of platinum ions into their electrically neutral form.
This caused the platinum to deposit into tiny metallic clumps on the surface of the liposomes over the site of each porphyrin.
Once each clump grew to about 500 metal atoms, the platinum catalyzed its own growth and formed large, branching sheets that spread over the surface of the liposomes. Finally, when the liposomes themselves aggregated, the platinum arranged into structures resembling nanoscale sheets of foam. Shelnutt’s team describes the new metal-processing strategy in the Jan. 21 issue of the Journal of the American Chemical Society.
By increasing the duration of light exposure or the concentration of porphyrin molecules in the liposome membranes, the Sandia team can increase the size of the platinum clumps, which leads to larger metallic foams. One potential application became apparent when the researchers irradiated the water-immersed structures and observed hydrogen emanating from the platinum foam. That same reaction could be used to make the hydrogen needed to power fuel cells.







