A portable sensor that borrows a trick from biotechnology could lead to rapid detectors of environmental uranium contamination, researchers say.
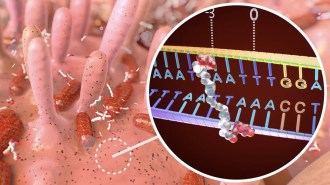
Yi Lu of the University of Illinois at Urbana-Champaign and his coworkers built a sensor containing catalytic DNA, which can split another DNA strand at a specific site. Most catalytic DNAs require a metal ion to function, and the uranyl ion triggers activity in the sensor. This ion is the most stable form of uranium in water and can be readily taken up in tissue.
The sensor is a solution containing the catalytic DNA along with its target DNA strand, which is attached to a fluorescent molecule. A so-called quencher sits next to the fluorescent molecule and normally keeps it turned off. When the researchers add uranyl ions to the solution, the catalytic DNA clips the target DNA, releasing the fluorescent molecule. Freed from the quencher, the molecule glows, signaling the presence of uranium.
In laboratory tests, the sensor detected as little as 11 parts per trillion of uranium in less than 2 minutes, the researchers report in the Feb. 13 Proceedings of the National Academy of Sciences. That amount is below the concentration considered toxic in drinking water.
The sensor’s performance compares favorably with that of large laboratory machines, Lu notes. An additional advantage is portability. “We want to fill in the gap for on-site, real-time detection,” he says.