Mapping aggression circuits in the brain
Scientists are studying what makes flies and mice lash out

FIGHTER FLY The fruit fly Drosophila (above) has attack-promoting nerve cells similar to some in mice and birds. Studying such cells might help scientists better understand human aggression.
Eye of Science/Science Source
Male mice in David Anderson’s Caltech laboratory are typical rodents. Mellow most of the time, they’ll defend their cages if provoked — chasing male intruders away. The mice will lunge and nip until one admits, paws up, social defeat. Rarely do they actually hurt each other.
But with the flick of a switch, Anderson’s team can convert an ordinary lab mouse into a vicious brute that won’t back down. Like Bruce Banner morphing into the Hulk, the mouse seems to have no choice but to let the monster spring forth, inflicting bite after bite on its cowering victim.
To draw out the animal’s natural aggression, scientists activate a small group of nerve cells, or neurons, identified by Anderson’s group and others, that act as a control center for aggressive behavior. Turning on those brain cells instantly increases a mouse’s appetite to fight.
During these confrontations, the scientists map the physical and chemical connections among the cells to track the neural roots of violent behavior. Such rodent scuffles may seem a far cry from, say, the aggression of a school bully or cold-blooded killer. But researchers think some of the same circuits in the brain may be involved.
The brain’s attack neurons reside in an area called the ventromedial hypothalamus, a region long linked to aggression in animals. Scientists began closing in on those neurons in 2011 in the process of discovering a way to transform docile mice into angry goons.
Since then, researchers have narrowed the list of neurons involved. Last May in Nature, Anderson’s team described a small group of neurons that specifically escalate aggression. Nirao Shah’s team at the University of California, San Francisco independently identified the same neurons.
“In 2011, we knew the neighborhood where these neurons lived, but we didn’t know the exact houses or streets in that neighborhood,” Anderson says. “We now know that.”
Story continues after video
|
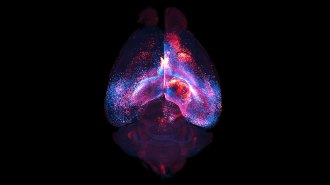
ROOTS OF VIOLENCE Tracking the networks of neurons that control aggression in mice and fruit flies could help us better understand how aggression is wired in the human brain. D. Lin; Unger et al. 2015; D. Anderson; ZEISS Microscopy; Davide; Eye of Science/Science Source; The CBI; J. Hirshfeld; Asahina et al. 2014. |
This aggression hub in the hypothalamus doesn’t work alone. It is part of a network of brain structures. From this neighborhood, where attack behavior is organized, each neuron connects to others throughout the brain to produce the features associated with fighting: a racing heart, increased metabolism and the muscle movements involved in charging or biting. By studying nerve signals coming into the hub and tracking those moving out, scientists aim to build a circuit diagram, showing how the brain puts the entire body into a fight state — or holds aggressive impulses in check.
Anderson calls this hub “our beachhead into the brain for studying aggression.”
Details of the map are still emerging. In some areas, neurons related to aggression differ in males and females. And neurons clustered in a second brain region of the mouse also induce an attack when activated, scientists reported February 3 in Cell Reports. These cells incite aggressive behavior in males and females alike.
Experiments in flies and birds reveal attack-promoting neurons in brain regions similar to those found in mice. Such studies of attack centers in animals promise a better understanding of how aggression is organized and produced in the brain: Is it distributed diffusely or controlled by a few key nodes? Because the human brain is wired pretty much the same as the mouse’s, the studies may lend insight into human aggression as well.
Bad neighborhoods
Occasional outbursts of aggression are common, even normal, in animals from birds to mice to humans. Faced with a genuine threat, most creatures will raise their voice or take physical action to fend off an attack.
Neurons that elicit attack behaviors sit in the hypothalamus — a region in the center of the brain. Formed by dozens of small clusters of cells, the hypothalamus governs many basic functions such as feeding, metabolism, body temperature, thirst, fatigue and sleep. It also regulates basic drives, such as mating and aggression.
In the 1920s, Walter Hess at the University of Zurich found that an electric burst to the hypothalamus could send cats into a fury, arching, hissing and clawing nearby objects. He was awarded a Nobel Prize in 1949 for his work. Studies in the 1980s confirmed that the hypothalamus is also a center for rage in rats. Still, the precise location of attack-promoting neurons, and their mode of action, remained a mystery. Without the tools to probe specific sets of cells in this hard-to-reach region, studies stalled.
In recent years, though, new technologies have made it possible to study individual brain cells in animals, so scientists could take a closer look at the role of the hypothalamus in aggression. One way to see where neurons are most active during a task is to track the activity of c-fos, a gene that produces proteins in response to neuronal activity.
With another technique, called optogenetics, researchers engineer individual rodent brain cells to make them controllable by light (SN: 1/30/10, p. 18). Scientists deliver a gene that encodes a light-sensitive protein into the cells of interest. The engineered neurons can then be activated or “turned on” to communicate with other neurons simply by shining colored laser light onto the cells. Scientists can stimulate or shut down specific cells and then observe the animal’s behavior.
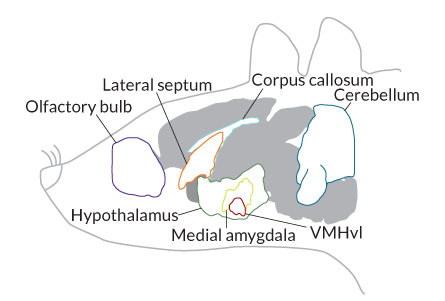
Using these techniques, Anderson’s group began probing cells in the hypothalamus five years ago. Dayu Lin, then a postdoctoral researcher in Anderson’s lab, quickly zeroed in on a small area that became active when male mice started to fight. This part of the hypothalamus, the ventrolateral area of the ventromedial hypothalamus or VMHvl, is made up of roughly 10,000 nerve cells.
“It’s not a lot of real estate in the brain, but apparently it is a very critical area,” says Lin, now at New York University. Her study, published in 2011 in Nature, also revealed a surprise: The small part of the hypothalamus linked with aggressive behavior is also associated with mating behaviors. In some instances, the same neurons activated during aggressive encounters appear to be activated when the animals mated.
Anderson’s team is looking for similar attack-mating connections in flies. In January 2014, his team reported in Cell on a tiny group of neurons that promote aggression in male fruit flies but don’t exist in females. These neurons are active when male flies jockey for position on a food dish or fight other males to win access to females. Because flies have a simple nervous system, activating only three to five neurons of their total of 100,000 brain cells sends them lunging at nearby targets. Anderson’s team is now investigating whether those neurons also play a role in mating, as they do in mice.
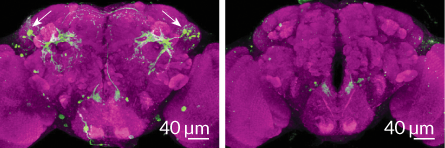
Whether VMHvl neurons are multitaskers, busily working to promote both sex and violence, or if they just happen to occupy the same general brain area, is not yet known. Even though these behaviors are opposing forms of social interaction, both are driven by chemical signals, called pheromones, and share common features such as sniffing and close physical contact.
“To an untrained eye, when you see two mice engaging in mating and fighting, it’s often difficult to tell the difference,” Anderson says.
Aggression and mating share another feature in animals: The behaviors are hard-wired from birth.
“The first time a male fly encounters a female fly, it will court her. The first time he encounters a male fly, he will engage in aggression with that fly,” Anderson says. The same holds true for mice: Aggression requires no formal training.
Given that the drive to express aggression is built into the brain, scientists say it’s no surprise to find that there’s a hub, or maybe several, directing behavior to make animals respond in the right way at the right time. Though researchers have yet to work out a flow chart, a chain of command might include a key command center with connections to nearby hubs and distant brain regions. The network would also connect to sensory neurons, which bring cues from the outside world (odors, sight, sound) to the hub to guide behavior, and to the brain’s motor regions to relay information needed to carry out actions.
From lover to fighter
Scientists are not looking for a single trigger to explain violent outbursts. Aggressive behavior, animal or human, is the outcome of not just the physical circuitry, but also brain chemistry, genes and a changing environment. Even within the VMHvl hub, there is much complexity. In this part of the hypothalamus different types of neurons do different jobs, relaying messages for mating, fighting and fear.
To resolve which neurons do what, Anderson’s group pored over the Allen Brain Atlas, an online catalog that maps gene activity in the adult mouse brain. In looking for genes that control the VMHv1 aggression hub, the scientists identified a group of cells marked by the presence of a receptor for the hormone estrogen. Follow-up studies showed that these neurons were most likely to be active when the animal was engaged in fighting.
The scientists then genetically modified mice by inserting light-sensitive proteins into those neurons. Surprisingly, by adjusting the intensity of light researchers could change the animal’s behavior. Activating the cells by turning the light up to high intensity prompts mild-mannered males to bite and claw nearby targets — even a mate or a castrated male, animals normally not perceived as threats. Turn down the light to low and instead of attacking targets, the mouse mounts them, a first step in mating behavior. Toggling the switch from low to high prompts animals to switch from lover to fighter in seconds.
Anderson says the findings, published last May in Nature, show that aggression neurons in the VMHvl control multiple phases of a social interaction, from sniffing and getting to know you, to touching and mating, to all-out attack. His group continues to alter activity in the cells in search of the control mechanism behind this fine-tuning, along with studying estrogen’s role in the mix.
Hormones are the body’s signaling molecules. Researchers have long known that the sex hormones — testosterone, estrogen and progesterone — have profound effects on the brain. They influence brain development in the young and memory in the old. Though viewed as the he-man hormone that controls aggression, testosterone gets converted into estrogen in the male brain. The enzyme aromatase handles this conversion.
In 2009, Shah’s group showed how estrogen “masculinizes” brain circuitry during development, predisposing boys to be boys by setting up scores of aromatase-producing neurons. Females have these neurons too, but in smaller numbers.
Since then, Shah’s group has probed the neural circuitry of male and female mice looking for differences that might explain sex-specific behavior. Even in mice, aggressive behavior differs dramatically between the sexes. Male mice forcefully stake out territory and constantly scuffle to maintain control. In the lab, a male mouse dropped into another male’s cage is immediately pummeled and chased away. Females do not perpetrate such attacks; they generally limit their aggression to protecting their young.
In addition, there are big differences in how males and females fight. Males, for example, typically attack other males by moving toward the flank region, or side. They may also box and wrestle. Females attack intruders by moving straight for the neck or the genital region. While both fight styles aim to ward off attacks, they employ different movements and strikes.
“It’s like one guy is using kung fu and the other animal is using tae kwon do,” Shah says. To study the neural pathways that control these sex-specific behaviors, Shah’s group has uncovered a handful of genes that are influenced by sex hormones in the brain. By turning off these genes, one by one, his group can tease apart a hormone’s influence on individual behaviors.
Through this process, Shah and colleagues identified a population of about 2,000 neurons linked to male aggression — roughly the same group of VMHvl cells identified by Anderson’s team. The findings, published in 2013 in Cell, showed that, along with estrogen receptors, most of the cells harbor progesterone receptors. Progesterone helps control sexual behavior and pregnancy in females, but is also produced in males. It’s not yet clear that receptors for either estrogen or progesterone play a direct role in controlling aggressive behavior, Shah says. For now, the receptors serve as a marker he can use to isolate and manipulate the cells.
Forging the path
In recent months, scientists have identified groups of aggression neurons inside the medial amygdala, a brain structure that processes fear and other emotion. Last September in Cell, Anderson’s group described a population of amygdala neurons that, when activated by optogenetic techniques in male mice, can either trigger attacks (at high levels of light) or produce mounting behavior (at lower light levels), just like the neurons in the hypothalamus.
Anderson says the amygdala neurons that promote attacks may be part of a circuit that feeds in — directly or indirectly — to the “attack” neurons in the VMHvl. His group is now searching for anatomical pathways between those structures.
In February, Shah’s team identified a group of aromatase-producing neurons in the amygdala. These cells are the first in the aggression circuit shown to influence both male and female aggression, Shah says. His group is working to identify connections going to and from this region to see if aggression-related neurons are wired into different circuits in females than in males.
As details of the aggression circuit emerge, scientists in these labs and others are designing experiments to determine how, or if, environmental cues feed into the circuit to help control, or resist, impulses to fight. In her NYU lab, Lin uses optogenetic tools to trace signals entering the middle of the VMHvl from a brain area called the lateral septum. In mice, the lateral septum is sandwiched between the cingulate cortex and the nucleus accumbens, near the front of the hypothalamus. The lateral septum has physical links to the hypothalamus, with projections leading directly to the VMHvl.
Studies in rodents show that damage to the lateral septum causes animals to become hyperaggressive. Because cells in this brain area produce inhibitory neurotransmitters that slow down neurons, scientists believe that, under normal circumstances, the structure serves to inhibit aggressive impulses.
“The lateral septum seems to be perfectly positioned to relay those environmental and experience-related cues to moderate the output of the social behavior,” Lin says.
As studies progress, questions about how the brain controls aggression may get back to even more basic questions, such as: How can people actively control their aggressive impulses? Or create a safer world?
Anderson says biological studies of violent behavior are often hampered by legal and social concerns. Could individuals be unfairly stigmatized if they are wired for violent tendencies? Would knowledge of aggression’s biological roots make it hard to convict the pathologically violent, who might claim, “My cells made me do it”?
“On the one hand, we’re terribly concerned about violence. Every time there’s another school shooting, people want to know why there’s an epidemic of violence in this country,” Anderson says. “At the same time, people are very reluctant to get to the biological roots of violence and aggression. But if we knew something more about it, we might be able to prevent some of these tragic events.”
This article appeared in the March 21, 2015 issue of Science News under the headline, “Aggression Avenue: Tracing how the brain is wired for violence.”