Model Growth: Simulations expose branching nature of polymer crystals
The intricate shapes of snowflakes have long fascinated scientists, but water isn’t the only fluid that freezes into elaborate crystal structures. Polymers and metal alloys do the same. Now, scientists in the United States and Hungary have uncovered previously unknown facets of the physics underlying such crystal growth.
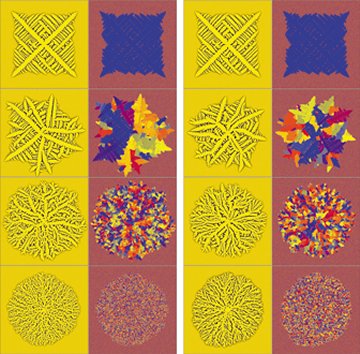
Using computer models, the researchers simulated a polymer liquid as it solidifies into a crystal. In one scenario, the researchers added increasing amounts of dirt particles, representing impurities, to the liquid and confirmed previous observations that the more dirt present, the more complex the resulting polymer crystal.
Normally, a polymer in solution begins solidifying by forming tiny needle-shaped crystals. These grow along one direction, eventually packing into a polycrystalline structure. However, when one of these needle crystals encounters an impurity, “it has to figure out how to incorporate the particle into its structure,” says James Warren of the National Institute for Standards and Technology in Gaithersburg, Md.
He and his colleagues at the Research Institute for Solid State Physics and Optics in Budapest found that in their model, the position and orientation of a particle can make the polycrystal growth veer in a new direction, creating a branch point. Therefore, the more dirt particles it encounters, the greater the randomness of the branching structure. The researchers dub polycrystals that follow these branching growth patterns “dizzy dendrites.”
Warren’s group also reports a surprising discovery: Dirt isn’t required for branching structures. When the team modeled a dirt-free solution and simulated it being chilled below its normal freezing point, the polycrystals that formed were as highly branched as were the dizzy dendrites produced from solutions with impurities.
Warren notes that many small crystals form independently in a polymer solution. Generally, a small crystal rotates to line up with a growing polycrystal before joining it. However, in a supercooled solution, the small crystals rotate so slowly that they become incorporated in whatever orientation they happen to be in when they encounter the polycrystal. The researchers describe their simulations in the September Nature Materials.
“The simulations employed in this paper are truly state of the art,” says Mark Asta, a computational materials scientist at Northwestern University in Evanston, Ill. Because a material’s microstructures determine its mechanical properties, the findings could help scientists design improved materials—for use in products ranging from plastic grocery bags to airplane wings—by controlling the way a material crystallizes, researchers contend.







