Like tiny locks, receptors in biology respond exclusively to the

specific molecules that serve as miniature keys. Many basic biological
processes, from sensing of odors to signaling between nerve and
muscle cells, rely on such lock-and-key combinations. Scientists trying
to understand these interactions often play locksmith, searching
for the right key to fit a particular receptor.
Now, using this biochemical machinery as a model for a variety
of applications, scientists are building the locks themselves. They
use a new technique called molecular imprinting. The researchers
etch a material to create molecule-size, carefully shaped pores that
amount to artificial receptors.
The shape and chemistry of these pores also enable them to
serve as enzymes, selective membranes, and specific detectors.
Researchers around the world are hoping that molecularly imprinted
materials can be used to separate chemicals for industry, detect biological
molecules, and develop biomedical implants.
“The field is exploding,” says Klaus Mosbach of Lund University in Sweden.
Improving techniques
A desire to improve the techniques available to purify chemical components of mixtures led
Mosbach to molecular imprinting. He had been working on affinity chromatography, a technique
in which researchers pack a long tube, or column, with a material that binds preferentially to a
particular compound. When a solution is forced through the column, the compound of interest
sticks to the material, and unwanted substances flow out the far end. Later, the desirable compound
can be washed from the packing material
and collected as it streams out of the column.
Mosbach took this technique a step further
by attaching enzymes to little polymer beads,
which he then used as column-packing material.
The bound enzymes promoted reactions among
chemicals in the solutions passing through.
This provided a practical way for pharmaceutical
companies to make large batches of drugs.
Instead of using “the power of microorganisms”
in bacterial cultures or yeast fermentations to
change steroids like cortisone into drugs like
prednisone, Mosbach says, the companies
“could add a steroid to one end of the column
and get product at the other end.”
Eventually, Mosbach and his group decided
to make the method more reliable under different
conditions by bypassing enzymes altogether.
By creating tiny cavities in a polymer, or
plastic, that mimic the active sites of enzymes,
the team essentially produces artificial enzymes, or “plastizymes,” as Mosbach calls them.
Self-assembly
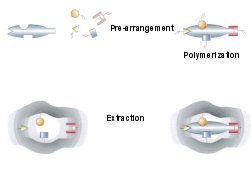
Researchers have succeeded in creating dozens of these polymers. Mosbach and his colleagues
manufacture the catalytic plastics via a method called self-assembly (SN: 3/1/97,
p. S14). For a given target molecule, they choose a variety of simple chemical units, or monomers,
that they predict will interact with the functional groups on the target. For example, if the
target has positively charged groups, then negatively charged monomers go into the mix. If it
has groups that will form hydrogen bonds, then the researchers choose other monomers that
can complete those bonds.
When mixed together, the monomers surround the target molecules in a specific arrangement
dictated by properties of both components and their interactions. The scientists then add
crosslinking polymers that solidify the material into a rigid gel. Finally, the researchers wash
out the original target molecules, which leave their imprints in the plastic.
These precisely shaped imprints are “cavities with antifunctionality–like the active site of an
enzyme,” Mosbach notes.
One of his group’s current projects is to develop plastizymes that can degrade toxic compounds.
Kenneth J. Shea of the University of California, Irvine compares this imprinting to making a
plaster mold of someone’s right hand. Not only does the final cast perfectly fit just one person’s
hand, but it matches the right hand and not the left.
The comparison is apt because Shea and his team are developing imprinted polymers for
separating so-called chiral compounds. Like a pair of hands, chiral molecules of a compound are
mirror-image versions of each other. Separating these closely related forms is difficult to do
chemically.
In some cases, drugs come in chiral forms that have very different properties–one may be
therapeutic while the other causes disastrous side effects. Shea and his colleagues have used
imprinted polymers to separate chiral forms of benzodiazepines, a class of drugs that serve as
sedatives and muscle relaxants. They report their latest findings in the Jan. 26 Journal of the American Chemical Society.
The imprints act like the natural benzodiazepine receptors, which are made of protein. The
plastic versions are “somewhat primitive compared to the best protein recognition sites, but
they’re manmade,” Shea says. “The material is more robust, unlike proteins, which are sensitive
to pH, heat, et cetera. You can put the polymers on a hot plate and not lose recognition sites.”
Good for screening
This hardiness makes imprinted polymers good for screening large numbers of compounds.
As part of their strategy to select new drugs, pharmaceutical companies test thousands of
chemicals to select the few that bind to a particular receptor and thus stimulate or block a biological
effect.
Acting as artificial receptors, imprinted polymers could also work as sensors. For example,
in one project Shea’s group is making polymers to detect brevetoxin, a fish-killing compound
produced by the algae that create red tides (SN: 9/27/97, p. 202). “It doesn’t respond to conventional
assays,” Shea says, so samples have to be shipped to distant specialized laboratories for
chemical identification.
His vision is to be able to detect brevetoxin with a method as simple as a home pregnancy
test. A strip coated with a molecularly imprinted polymer and a fluorescent dye could serve as
an on-the-spot indicator. Dip the strip in the water, and if it lights up, then the toxin is present.
Shea and his colleagues have also crafted molecularly imprinted polymers into films that act
as semipermeable membranes. Like natural cell membranes, the polymer films let certain substances
pass through but blocks others. The researchers found that their polymers could distin guish between very similar compounds. One film that they made permits adenine–one of the
four bases that carries the code in DNA molecules–to pass through but blocks the other three
bases.
The ability of imprinted polymers to mimic the human body’s receptors isn’t lost on materials
scientist Buddy D. Ratner of the University of Washington in Seattle. He and his colleagues
are creating imprinted polymers for biomedical implants.
“The materials that are in wide use in medicine, as far as the body is concerned, [are] all foreign
materials,” Ratner says. The body has a way of dealing with those interlopers: “It puts a
wall around them,” he explains, and keeps up a low level of inflammation.
Ratner predicts that molecularly imprinted materials could be integrated into a person’s
body instead of being isolated from it. He and a team of biologists, engineers, and materials scientists
are trying to identify molecules that contribute to wound healing.
An implant made of an imprinted polymer could attract proteins that would encourage the
body to integrate the implant with the natural tissue. The group demonstrated that artificial
receptors for large proteins could indeed be imprinted into a polymer. They have successfully
created pores for bovine serum albumin, immunoglobulin G, and fibrinogen, which are typical
of proteins that mediate the body’s response to foreign objects.
Technique extended
Companies use catalysts to speed many chemical processes. To use molecular imprinting,
they would need imprinted materials that are tougher than polymers because reactions often
take place under high temperatures and harsh conditions. Alexander Katz of the University of
California, Berkeley and Mark E. Davis of the California Institute of Technology in Pasadena have
extended the imprinting technique to silica, the main component of glass.
To the naked eye, imprinted silica looks the same as an ordinary piece of glass, says Katz. In
the Jan. 20 Nature, he and Davis describe their imprinting process, which constructs a silica network
around the template molecules by the sol-gel method, in which the researchers apply a
solution of silicon compounds and let the solvent evaporate (SN: 1/25/97, p. 56).
Their study broke new ground on two fronts, says Katz. They measured the number and size
of the cavities available to bind the target molecules. “This is the first time in the history of the
imprinting field that we can directly measure the pore volume of the material,” says Katz.
Also, because the material is inorganic, the researchers can use nuclear magnetic resonance
spectroscopy to track the binding of organic molecules. A polymer would create so much background
noise that the molecules would be impossible to see using that technique. These
advances will help researchers characterize the imprinted materials, he says.
“We hope to learn from biological systems to make catalysts that nature never had the need
to make,” says Katz.
Early stages
Research into imprinted materials is still in its early stages. Scientists have some critical
issues to resolve before the materials can become practical.
For one, they will have to increase the density of sites on the polymers, says Shea. Because
the polymers have to be rigid, a lot of the volume is taken up by crosslinkers that connect the
floppy molecules together.
The low concentration of sites is also due to the imperfect synthesis method. They “just
throw it all together, and let ‘er rip,” Shea notes. This approach creates many good recognition
sites, but also a fair number of bad ones.
Molecularly imprinted materials have a long way to go to rival natural proteins, which
arrived at their present design through millions of years of evolution. A one-step polymer or
sol-gel synthesis can’t capture all of the subtleties of an enzyme or antibody. Scientists, though,
are giving it their best shot.






