New brain cell growth restores function
Regeneration helps repair learning and memory after injury in mice
Newborn nerve cells may help heal the brain after a traumatic injury.
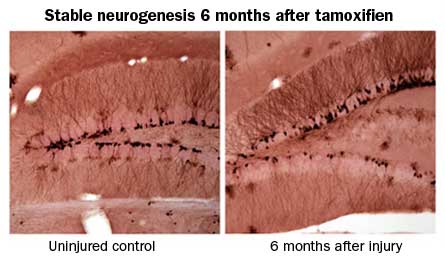
In a study in mice, blocking the birth of new neurons hindered the mice’s ability to learn and remember a water maze after a brain injury, researchers from the University of Texas Southwestern Medical Center at Dallas report in the March 30 Journal of Neuroscience. The finding could help settle a debate about what new nerve cells do for the brain and may eventually change the way brain-injured patients are treated.
Although scientists have known for a decade that adult brains can make new neurons in two parts of the brain, the role of the newborn cells has not been clear. Some scientists thought that, in adults, neurogenesis, as researchers call the process of generating new nerve cells, may be a leftover from building a new brain during development and has no affect on the adult brain at all. Others have evidence that the new wiring that hooks up new brain cells sometimes gets tangled and may lead to seizures after a brain injury or in epilepsy. Many researchers have suspected that making new cells is good for the brain, but data to definitely settle the claim has been lacking.
The new study suggests that newborn neurons made in the hippocampus — an important learning and memory center in the brain — are beneficial, at least in aiding recovery after traumatic brain injuries. “It’s clear they are doing something, and that that something aids recovery,” says Jack Parent, a neurologist and neuroscientist at the University of Michigan Medical Center.
To discover what role, if any, neurogenesis plays after a brain injury, the Texas team genetically labeled newborn cells in the hippocampi of mice. The researchers found that traumatic brain injury stimulates birth of more brain cells than usual. Using another genetic technique, the researchers blocked neurogenesis at the time of injury (to the brain’s cortex) in some mice. Mice that couldn’t make new brain cells didn’t recover the ability to learn a water maze after brain injury as effectively as brain-injured mice that could generate new neurons. The result indicates that newborn neurons are needed for learning tasks that involve the hippocampus, such as learning to find a submerged platform in a water maze. Parent says its still an open question whether the new cells help directly with learning and memory or indirectly by reducing anxiety, which can also affect performance in water-maze task.
But new cell growth has no role in making fear or motor memories, which don’t require the hippocampus. Mice in which new neuron production was blocked after brain injury were able to learn that a sound preceded a mild foot shock and how to cling to a rotating bar about as well as brain-injured mice with neurogenesis.
“This suggests that if you get in the way of neurogenesis in a big way, it’s not good,” says Patrick Kochanek, an intensive care physician and brain injury expert at the University of Pittsburgh School of Medicine. The worry for doctors now is whether some standard treatments for brain injury, such as sedation, might slow down neurogenesis and hinder recovery, he says. The new work might also suggest that using techniques that safely stimulate even more neurogenesis could make recovery stronger.
Parent cautions against trying to make too many new neurons, however. His research suggests that some newborn brain cells wire into the wrong places and can lead to seizures. So the trick would be in controlling the new cells once they are born. “You need more neurons, but you need them in the right place,” he says.
Learning whether more neurogenesis means more recovery after a brain injury and discovering which molecules are responsible for the healing are next on the Texas team’s agenda, says study coauthor Steven Kernie, a developmental neurobiologist at UT Southwestern.







