Two centuries after Englishman William Hyde Wollaston first isolated the element palladium, researchers have now uncovered fundamental new information about the material.
Palladium–a soft, gray-white metal that resembles platinum–is a component in dental alloys, jewelry, and electrical contacts. It also serves as an important catalyst in countless chemical reactions, including those that occur in automobiles’ catalytic converters. Fundamental reactions widely used to produce pharmaceuticals, such as reactions that add hydrogen atoms to molecules, also rely on palladium catalysts.
With the help of a newly synthesized palladium compound, a team of Japanese researchers has uncovered chemical information that they say is important to understanding and designing such catalysts. The finding is reported in the Jan. 11 Science by Shigeru Shimada and his colleagues at the National Institute of Advanced Industrial Science and Technology in Tsukuba, Japan.
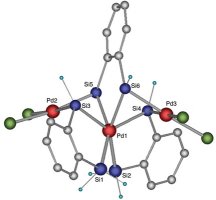
The new compound contains a palladium atom bonded to surrounding atoms in a way that scientists hadn’t observed previously. This novel arrangement seems to change the palladium atom’s so-called oxidation state, or oxidation number.
An atom’s oxidation state defines its capacity to donate, accept, or share electrons when it bonds to other atoms. It’s a concept familiar to generations of chemistry students and fundamental to scientists’ understanding of how chemical reactions work.
“Half the reactions in nature deal with oxidation state,” says chemist Robert H. Crabtree of Yale University.
A particular element can have different oxidation states. The new structure’s arrangement suggests that its palladium atoms are in the highest oxidation state ever observed for the element, says
Shimada.
The researchers created their compound by heating molecules containing carbon, hydrogen, silicon, phosphorus, and palladium for 2 days in the solvent toluene. Three of these molecules, each containing a palladium atom with the oxidation number 2, combine to form the product, says Shimada. The resulting red, crystalline compound has a central palladium atom bonded to six silicon atoms, giving it an apparent oxidation number of 6, the researchers report.
They base their conclusion on X-ray analysis, nuclear magnetic spectroscopy, and infrared spectroscopy.
There is another interpretation of the data, Shimada and his colleagues acknowledge. Some silicon atoms in the structure are close enough together that there could be some partial bonding between them. Shimada notes that this interpretation would yield an unremarkable oxidation number of 2 for the new compound. His team plans to do calculations to attempt to settle the issue.
Regardless of whether the oxidation state turns out to be 2 or 6, Crabtree says, the new compound has a unique structure and gives interesting information about the nature of palladium bonding.
Shimada expects the results to deepen chemists’ understanding of catalytic processes in general and also lead to new palladium-containing catalysts.






