First, let’s clear the air: Nicotine invites addiction, and it employs a delivery device that’s been killing people for centuries. But let’s also be honest: Nicotine has some attractive qualities. Smokers use it to calm jitters or perk themselves up. It’s a common (if ill-advised) tool for weight control. Nicotine lowers the risk and eases the symptoms of Parkinson’s disease. Patients with mental illness have high rates of tobacco use, partly because nicotine helps quiet the mind.

Because of these talents—along with nicotine’s intense grip on the brain—scientists have sought compounds that can deliver the good without the harm. Now, almost 20 years after the quest began, the research has come a long way, baby. A number of experimental drugs—molecules reduced to mere shadows of nicotine—show signs of being able to exploit nicotine’s power to compensate for the defects in an ailing brain.
Such drugs may offer new therapies for diseases that now have few treatment options—boosting cognition in patients with Alzheimer’s disease, calming hyperactivity, relieving pain or treating mental illness. Similar drugs are also in early testing for Parkinson’s disease, inflammation and even obesity.
“We’re very blessed that nature gave us nicotine,” says Donald deBethizy, chief executive officer of North Carolina–based Targacept Inc., which broke off from tobacco giant R.J. Reynolds in 2000. Few other compounds, deBethizy says, affect the brain at such a basic neurological level, with so much power to control chemicals that ferry signals from one brain cell to another.
Most common neurological diseases reflect problems with one of those messenger chemicals, or neurotransmitters. Drugs for such diseases usually target the activity of one neurotransmitter at a time. But nicotine is a volume knob for many at once. And that could make nicotine-mimicking drugs both potent and hazardous, affecting brain function so fundamentally as to cause worrisome side effects. In fact, the first modern drug approved that acts directly on the brain’s nicotine relay system—the smoking-cessation drug varenicline—has been tarnished by anecdotal reports of nightmares and suicidal thoughts in people taking the drug. How much those problems can be blamed on the drug’s action remains unclear, but it’s hard to find a researcher in the field who hasn’t thought long and hard about side effects.
“The issue is, as with all drugs, they have pluses and minuses,” says Allan Collins of the University of Colorado at Boulder. “Finding compounds that have more pluses than minuses has been the problem.” But, he adds, “I’m more optimistic than I have been in the past.”
Beginning with a bad reputation
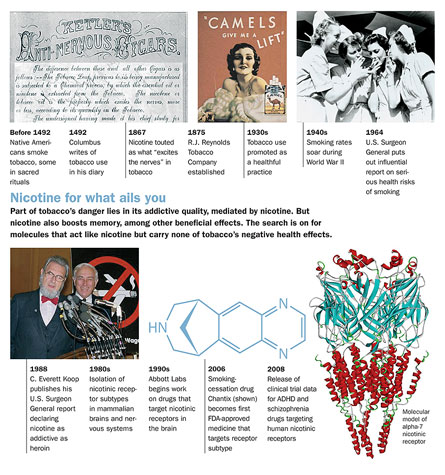
In this case, the past dates back to the late 1980s, when researchers began to realize the breadth of the brain’s inventory of nicotinic receptors. Receptors are like docking stations, allowing molecules to attach to a cell and set a chain of events in motion. Usually each molecule lodges in a specific receptor on a cell, like a garage that houses only one make and model of vehicle. Once the molecular car is parked, a lot of running around occurs inside the cellular house. In the case of nicotine, one molecule pulls into lots of garages.
In a normal, tobacco-free existence, these receptors aren’t sitting around waiting for nicotine. They are made for acetylcholine, a powerful neurotransmitter associated in the brain with attention, learning and memory. Nicotine just happens to be a sometime impostor for acetylcholine in smokers. “Nicotine is a sloppy drug that acts at all these receptors,” says Martin Sarter of the University of Michigan in Ann Arbor. Also, nicotinic receptors adapt to chronic exposure over time. The changes differ depending on the type of receptor and where it lies in the brain. Such changes are thought to explain why smokers lose sensitivity to nicotine, eventually requiring higher doses to achieve the same effect.
As they identified more and more receptor subtypes, scientists began to wonder how the brain would respond to a nicotine-like molecule that could activate just one type of acetylcholine receptor. For 15 years or so, not many people cared to know. Aside from isolated academic exploration at universities, the vast majority of commercial drug pursuit came from the R.J. Reynolds team that would become Targacept, as well as the Illinois-based firm Abbott Laboratories. It seemed that nicotinic receptors’ pedigree presented a public relations headache. Even if the research could produce great new drugs for desperate needs—consider the prediction that more than 8 million North Americans will develop Alzheimer’s by 2050—pharmaceutical executives feared that nicotinic drugs would have a questionable image. (Indeed, the corporate public affairs office at Abbott said the company would not contribute to this story.)
“It was very difficult in the beginning,” says Stephen Arneric, who worked on nicotinic drugs at Abbott during the 1990s, but has since joined the biotech startup Neuromed. “When the program was started at Abbott, the first question out of senior management was, ‘Merck isn’t doing it, why should we?’” Arneric and his colleagues even launched a quiet, so far unsuccessful, campaign to change the name of the receptors, removing the regrettable name “nicotinic” to make the research more palatable.
“The demonization of nicotine is what we’re all up against,” deBethizy says. However, unlike the lonely years of the 1990s, it’s now difficult to find a major drug company not trying to capitalize on nicotinic receptors.
Receptor by receptor
Chemists have produced molecules with selectivity for only one particular receptor type at a time. The biggest players remain Abbott and Targacept. Generally, drugs in development target two particular subtypes of nicotinic receptors, known in shorthand as alpha-4-beta-2 and alpha-7. Other types of nicotinic receptors are scattered throughout the brain, in muscle and in cardiac tissue. But in the brain these two appear to be common and best suited for disease treatment. Farthest along in testing are drugs for Alzheimer’s disease, depression, pain and attention-deficit/hyperactivity disorder, or ADHD.
In 2006, Targacept researchers released the results of a study of a nicotine-like drug designed to improve cognition in people with Alzheimer’s disease. The drug is designed to bind only to the alpha-4-beta-2 receptors in the brain and boost other neurotransmitters. In collaboration with AstraZeneca Pharmaceuticals, the company completed a randomized trial involving 193 patients with memory impairment (though not dementia) related to Alzheimer’s or schizophrenia. After taking the drug for 16 weeks, patients showed improvements on tests of attention and memory, the researchers reported during an Alzheimer’s research meeting the same year. But in September, the company issued a press release calling results of a larger study “inconclusive.”
A separate Targacept drug, this one for depression, is also working its way through the testing process. In 2007, at a meeting of the British Association for Psychopharmacology, the company described a trial involving 184 volunteers who had not responded well to first-line antidepressant drugs. The results of this study were encouraging enough to expand trials, though the true effectiveness of the drug—as with all nicotinic compounds—won’t be known until further studies are completed.
Also, a partnership of two companies, Abbott and the Denmark-based NeuroSearch, announced this year the results of tests on a drug for ADHD. That study, described in May during the annual meeting of the American Psychiatric Association, involved 221 adults with ADHD. After taking the drug for four weeks, volunteers reported an increase in work productivity and a reduction in absenteeism. Based on these results, the companies plan to move the drug into further testing. In a twist that speaks to the broad influence of nicotinic receptors in the brain, the same drug also appears to be a candidate for pain control.
Not all testing falls under the domain of pharmaceutical corporations. In August, a research team led by scientists from the University of Colorado Denver published the results of a study testing a nicotinic compound for schizophrenia, a disease that has been the subject of a decades-long search for new treatments. Upwards of 85 percent of people with schizophrenia smoke, largely because many patients feel a greater mental clarity when they light up. Writing in the August American Journal of Psychiatry, the research team described the results of tests in 31 people who received two different doses of the experimental medication. The molecule targets the alpha-7 receptors in the brain. The results were heartening, though not dramatic, and the usefulness of the drug is still unknown.
Strong medicine
In most of these early drug trials, complaints have been similar to those felt by many a teenager sneaking his first cigarette—nausea, diarrhea, dizziness. Nonetheless, side effects are still among the biggest concerns. Everyone in the field took note in February when the U.S. Food and Drug Administration hastily called a news conference to announce a public health advisory for varenicline, the antismoking drug made by Pfizer under the trade name Chantix. The FDA warned of mood changes, suicidal thoughts and suicide. “Clearly these are very concerning findings for this product which is being widely used,” the FDA’s Bob Rappaport told reporters.
Until these reports began to surface, the approval of Chantix had been a bellwether in the race for nicotinic drugs: a medicine that had entered the market and shown record success. Smokers taking Chantix have a much better track record of giving up cigarettes than those trying other methods. Animal studies suggest the drug might one day have a role in treating alcohol addiction. The side effects remain unexplained.
“They may indicate the heterogeneity of the human brain and the condition,” says Roger Papke of the University of Florida in Gainesville, who has long studied nicotinic receptors. As with other illnesses, each person may have variations in receptors that alter his or her response to the drug.
Varenicline binds to nicotinic receptors, interfering with the mechanism most strongly associated with addiction, with the idea of still allowing the brain enough reward to ease withdrawal symptoms. Whether the molecule in its current form binds too many types of acetylcholine receptors, whether it activates other neurological systems or whether suicide risk is simply elevated among people giving up cigarettes is unknown.
“I don’t think they’re going to see it in all drugs,” Arneric, the former Abbott scientist, says about these types of side effects. “If you look at nicotine itself, it doesn’t have those side effects.”
An extra dose
Developers of nicotinic drugs have had to make one other consideration: 20 percent of the adult U.S. population smokes, and will administer their own nicotine on top of whatever drug they may receive for an illness. In doing so, they will be stimulating nicotinic receptors in the blunt, old-fashioned way along with a treatment for memory or pain. No one can say with certainty what that means.
Then there’s the property of nicotine everyone knows—addiction. Can a medicine that works like nicotine, even if it isn’t really nicotine, completely rid itself of nicotine’s most notorious quality? “The exact basis for the addiction to smoking—that is more complicated than it seems,” says University of Michigan’s Sarter. “Is it possible that these selective compounds produce an addictive component? I don’t think we know at this point. Personally, I don’t think this is the case.”
DeBethizy of Targacept—who spent years as an R.J. Reynolds scientist—believes that addiction to nicotine has much to do with binding to another type of receptor, alpha-6. “We’ve decided that the best way to make nicotinic drugs is to develop selective compounds,” he says. “Steer away from a form of alpha-6, which is involved in addiction.” Though it’s still too early to draw conclusions, he says that so far the nicotinic drugs haven’t shown signs of causing cravings or dependence.
Which means, for the first time in human history, drugs may come along that have a nicotine-like relationship with the brain in a way that has nothing to do with addiction. “There will be some good new medicines,” Arneric predicts. If that’s true, a drug that has been an accomplice in millions of deaths may finally have a chance to help make some lives better.
Laura Beil is a freelance science writer based in Texas.
Nicotine for what ails you
Click on the image below to view an enlarged timeline on Nicotine and its discovered effects.