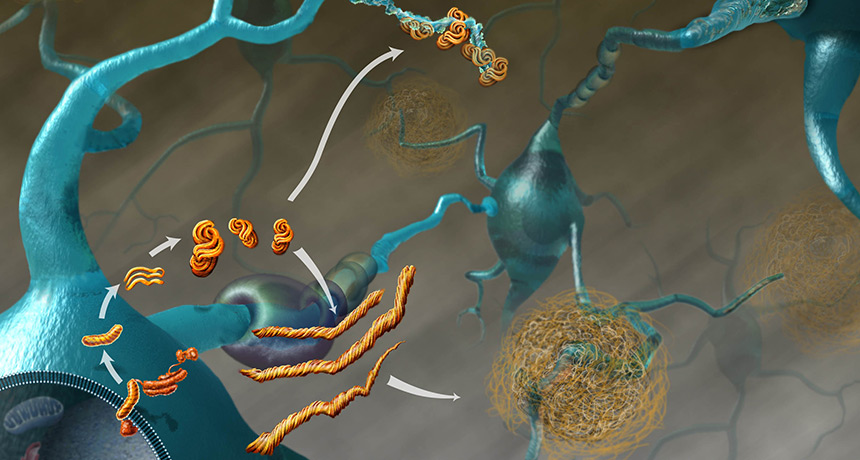
BRAIN DRAIN Prion diseases, such as kuru and mad cow disease, result when a normal brain protein (lower left) morphs into a disease-causing form and converts its normal counterparts. The twisted proteins form clumps (upper center) or strands that cluster in large plaques (lower right) that kill brain cells.
Nicolle Rager Fuller
Ending cannibalism stopped a deadly brain-wasting disease called kuru. But evolution already had devised a cure for the prion disease, a new study shows.
Some of the Fore people of Papua New Guinea have a naturally occurring variant of a brain protein that prevents kuru and related diseases, researchers report online June 10 in Nature. “We’ve never seen anything before that is completely protective,” says study coauthor John Collinge, a neurologist and molecular biologist at University College London. “It just switches off the disease.”
Understanding how the variant protects against prion diseases may also give new insights into Alzheimer’s, Parkinson’s and many other neurological diseases caused by twisted forms of normal brain proteins, he says.
Kuru results when a normal brain protein called PrP gets twisted into a disease-causing, or prion, form. The rogue protein goes on to corrupt the normal form of the protein into the disease form. The twisted proteins clump together and kill brain cells, eventually killing the person.
Other prion diseases include bovine spongiform encephalopathy — better known as mad cow disease — in cattle, scrapie in sheep and Creutzfeldt-Jakob disease, which sporadically strikes about one in a million humans. People can contract variant Creutzfeldt-Jakob disease, or vCJD, from eating beef tainted with mad cow disease. Each of the diseases contorts the PrP protein differently, producing different symptoms.
The prion disease kuru first appeared in the 1890s, cultural anthropologists Shirley Lindenbaum of City University of New York and the late Robert Glasse learned from interviewing Fore people. It spread when Fore women and children ate kin during funeral rituals. By the 1950s, kuru became a leading killer of the Fore, claiming up to 2 percent of the population annually in each village. Ritual cannibalism ended in the late 1950s after intervention by the Australian government and kuru cases dwindled, Lindenbaum says. The last known death from kuru was in 2009.
Natural selection was already at work to wipe out kuru. Sometime in the distant past, ancestors of the Fore had developed a genetic variant that changes the 127th amino acid in the chain that makes up the PrP protein from glycine to valine, a change known as V127, Collinge and colleagues discovered. About 7 percent of the Fore people now have the V127 variant.
The researchers genetically engineered mice to make human versions of PrP in their brains. Some mice made only normal PrP. Others made both normal PrP and V127, and some made only the V127 version.
Mice with the protective variant don’t get kuru or Creutzfeldt-Jakob disease when injected with the infectious prions that cause those diseases, the researchers discovered. Mice that make both normal and V127 versions of PrP don’t die of vCJD, but do have some clumps of prions in their brains and about 10 percent develop symptoms. Mice making only V127 are completely protected against the disease.
The results indicate that V127 not only resists being converted into prions itself but also somehow protects the normal form of the protein from being corrupted, Glenn Telling, a prion researcher at Colorado State University in Fort Collins, writes in an accompanying commentary in Nature.
The researchers don’t yet know how having V127 prevents corruption. But “it must really throw a spanner into the works in terms of prion propagation,” Collinge says.
Even if cannibalism had continued among the Fore, the protective variant may have eventually stopped the kuru epidemic; anyone who didn’t have it would have been wiped out by the disease, Collinge says. Eventually those immune to kuru because of the protective variant would have repopulated the villages.






