Heavy water is not just heavier. Swapping each H in H2O with a D — hydrogen’s isotope deuterium — changes many of water’s properties. Heavy water is poisonous, and its freezing point is 4° Celsius, instead of 0°. Those differences may reveal that quantum effects rule in ordinary water, researchers have now found.
The results, reported in an upcoming Physical Review Letters, could shed light on quantum theory’s relevance for ordinary water, which is the medium for most of the action inside living cells. The work could also help explain some controversial findings on how biological molecules behave in water.
Quantum objects, such as atomic nuclei, have properties of both waves and particles. Quantum effects aren’t usually manifest to the naked eye, but in this case they may be responsible for some of water’s unusual features. “The quantum effect in water is abundantly obvious,” says Alan Soper of the Rutherford Appleton Laboratory in Didcot, England.
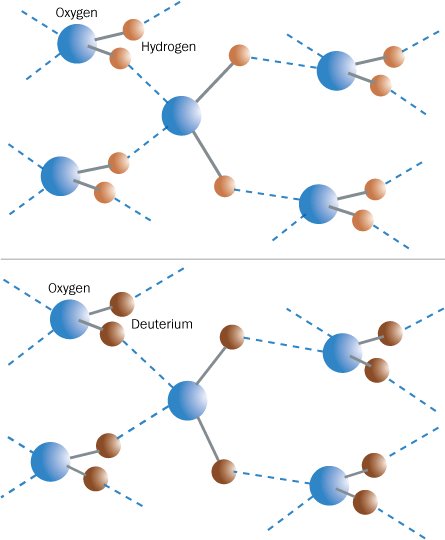
Working with Chris Benmore of the Argonne National Laboratory in Illinois, Soper has found that, in the liquid state, the distance between oxygen and deuterium in a D2O molecule is three percent shorter than the distance between oxygen and hydrogen in an H2O molecule. Conversely, hydrogen bonds — relatively weak bonds connecting the oxygen in one molecule with the hydrogen or the deuterium in another — are four percent longer in heavy water than in light. These differences are less than one percent in water vapor, where the molecules are isolated.
“A four percent change in bond length is quite a bit,” comments Michael Rübhausen of the University of Hamburg in Germany.
The researchers probed the distances via beams of X-rays and beams of neutrons, and ran computer simulations to help interpret the data.
The deuterium nucleus, which contains a neutron in addition to the usual single proton, is heavier than the hydrogen nucleus. That makes deuterium nuclei behave more like classical, as opposed to quantum, objects, so that their positions in space suffer less from the quantum uncertainty that “smears out” a proton’s location. “The heavier the particle, the more classical it behaves,” Soper says.
Deuterium nuclei’s more classical nature makes them stick closer to the oxygen nuclei they’re bound to within a heavy-water molecule, Soper says. On the other hand, an oxygen atom from a nearby heavy-water molecule exerts a weaker pull on the deuterium. As a result, the oxygen-deuterium bond between the molecules is longer than the oxygen-hydrogen bonds joining molecules in light water.
Water is a remarkable liquid — for example it has unusually high surface tension and it becomes less dense when it freezes. Quantum physics, through its effects on the hydrogen bonds, could be playing a significant role in water’s weirdness, Soper says. “Probably all the properties of water are affected by the hydrogen-bond length.”
Rübhausen says the difference in bond lengths could help explain some surprising results he and his collaborators reported last year. His team was comparing RNA made with ordinary organic molecules to RNA made of those molecules’ mirror images. Their goal was to shed light on why life always uses one type of molecule rather than the other.
Chemically, the molecules and their mirror images should be identical. But the researchers found small differences in the energy it takes to excite electrons in the two types of RNA — but only when the RNA molecules were suspended in ordinary water. When the researchers repeated the experiment in heavy water, the differences disappeared.
Bond lengths affect the electrostatic forces around water molecules, Rübhausen says, which in turn change the energy of electrons in a nearby RNA molecule. So the different bond lengths in heavy or ordinary water could somehow end up masking or enhancing the energy differences in the two types of RNA, Rübhausen speculates.







