Target: Celiac Disease
Therapies aimed to complement or replace the gluten-free diet
Not many kids can imagine a world without cereal, pizza, or cookies. But these are just a few of the foods that Stanford University biochemist Chaitan Khosla has had to teach his 6-year-old son to avoid. The boy has celiac disease, an inherited immune disorder, and it has sentenced him, for life, to refrain from eating anything that contains wheat gluten or similar proteins in barley and rye. In the boy’s small intestine, those grain components would trigger a chain of events that can cause bloating, diarrhea, and malnutrition. Without the restricted diet, the condition can lead to fatigue, migraines, dermatitis, anemia, and osteoporosis.

Khosla’s son isn’t alone in enduring his gluten-free lifestyle. Although celiac disease was considered rare in the United States a decade ago, recent tallies indicate that it may affect as many as 1 in 150 people, or 2 million in all. Many people with the condition aren’t properly diagnosed and suffer unexplained symptoms and potentially grave complications.
Before an astute pediatrician diagnosed celiac disease in Khosla’s son, the researcher had never heard of the disease. Neither had his wife, although she’d had gastrointestinal and skin problems for years. “She was one of the hundreds of thousands of misdiagnosed [people] out there,” Khosla says.
People in whom the disease is recognized must give up not only most grain-based foods but also soups, sauces, canned foods, and hundreds of other items.
Manufacturers frequently add gluten to those processed foods, and they charge hefty premiums for products prepared instead with rice proteins or other innocuous additives.
After sugar, Khosla says, gluten is the second most prevalent food substance in Western civilization. At a typical supermarket, “chances are you’re not going to be able to fill a grocery bag with gluten-free products,” he notes. Raising a young child to be accordingly vigilant about what he or she eats is “gut-wrenching,” Khosla says. If the pun is intended, his solemn tone doesn’t betray it.
With the aim of releasing his son, wife, and other people from their strict, lifelong diets, Khosla and other scientists have turned their attention to therapeutic alternatives that could short-circuit the disease’s development. The right drug might block the degeneration of the intestinal lining–the hallmark of celiac disease.
The researchers’ latest findings offer new insights to the disease’s biological mechanisms and an unprecedented understanding of at least three potential molecular targets for drugs.
The challenge ahead is to develop medications that can effectively strike these targets in the body. Although the trigger for symptoms comes from the diet, celiac disease–which is also called celiac (or coeliac) sprue, and gluten intolerance–is not an allergy. It’s more similar to complex immunological disorders such as multiple sclerosis, insulin-dependent diabetes, and rheumatoid arthritis, for which effective drugs have proven difficult to develop.
Even if scientists can devise a drug for celiac disease, it might complement the gluten-free diet rather than fully supplant it. Nevertheless, Khosla maintains hope that a prescription pill for celiac disease will exist by the time his son goes to college, 11 years from now.
Taking aim
Because gluten is a complex protein, normal digestion doesn’t completely break it down. Surviving pieces called peptides come in contact with the lining of the small intestine and the molecules of the immune system there. Whether that molecular encounter results in the immune overreaction at the heart of celiac disease depends on the type of immune molecules present.
|
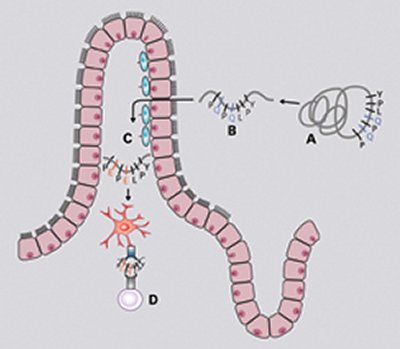
CELIAC LINKS. Digestion breaks down gluten (A) into peptides (B) that are absorbed by the small intestine. There, the enzyme tissue transglutaminase (C) modifies certain peptides, which are picked up by dendritic cells and, in people with celiac disease, stimulate immune cells (D) to attack the intestine.
|
Nearly all people with the disease have one of two immune-molecule types: About 90 percent carry so-called DQ2 molecules, and most of the rest carry DQ8 molecules. Genetics determines whether a person has either or both of these disease-associated molecules. For reasons not yet understood, a few people develop celiac disease despite having neither DQ2 nor DQ8 molecules, and not all people with the molecules develop the disease.
Early in both normal digestion and the pathological cascade that marks celiac disease, an enzyme called tissue transglutaminase (tTGase) alters gluten peptides.
When these altered peptides encounter DQ2 or DQ8 molecules in the intestine, they form molecular complexes that activate immune cells called T cells, which then mount an attack on the intestinal lining.
Under the assault, that stretch of the digestive tract becomes inflamed and loses the fingerlike projections, or villi, that normally provide a vast surface area for absorbing digested nutrients. Many of the symptoms of celiac disease, including malnutrition and anemia, develop in response to the small intestine’s reduced effectiveness in absorbing nutrients.
To forestall this cascade from occurring when a person with the disease ingests gluten, a drug could potentially dismantle all gluten peptides into their individual amino acids. That approach essentially treats gluten as a pathogen, Khosla says, and aims to detoxify the protein before it can cause trouble. Since the feat would probably require some sort of peptide-destroying enzyme, or peptidase, researchers refer to this approach as enzymatic therapy.
Two alternative approaches to blocking gluten’s toxicity would inhibit the activation of T cells by breaking other links in the pathological chain of events.
A drug could bind up tTGase, or another might fuse directly to DQ2 or DQ8 and take them out of the action.
If a single therapy can’t break a link in the chain, the best treatment might be a drug combination that weakens several links. A pill could theoretically deliver both enzymatic therapy to dismantle gluten peptides and compounds that inhibit tTGase, the disease-linked DQ molecules, or both, says Frits Koning of Leiden University Medical Center in the Netherlands.
Khosla and his colleagues have recently made progress on both these fronts. To figure out which peptides are most important in celiac disease, he, immunologist Ludvig M. Sollid of the University of Oslo, and other researchers tested peptides left intact after the team exposed gluten to mammalian digestive fluids. Among the digestion-resistant products, they discovered a peptide of 33 amino acids that seems to be a major source of the problem.
The researchers recognized that, given the order of its amino acids, this large peptide contains half a dozen locations at which it can interact with tTGase and DQ2. That may give it “exceptional toxic potency” in celiac disease, the researchers suggested in the Sept. 27, 2002 Science.
Khosla and his colleagues then sought a way to neutralize the newfound peptide. In experiments in rats’ small intestines, the scientists determined that a peptidase enzyme from the bacterium Flavobacterium meningosepticum rapidly breaks down the peptide. In the October 2002 American Journal of Physiology, they suggested that the bacterial peptidase could lead to an enzymatic treatment for celiac disease.
Looking also for an inhibitory approach to therapy, Khosla and several of his colleagues are investigating a way to bind up tTGase with an altered version of the recently discovered 33–amino acid peptide. Their artificial peptide differs by only one amino acid from the natural one, but it binds tightly to and thereby disables tTGase, the researchers report in the March Chemistry and Biology. That suggests that the artificial peptide could act as a tTGase inhibitor in the body, Khosla says.
Khosla’s recent publications are “certainly the most significant immunological findings” on celiac disease in years, says immunologist Allan Mowat of the University of Glasgow in Scotland. Nevertheless, Mowat says in the April 12 Lancet, barriers remain before the new insights can be turned into therapies.
One barrier facing the enzymatic approach, he says, is the difficulty of formulating a pill so that it releases a peptidase at just the right time. The enzyme could stay inside an acid-resistant coating as it passed through the stomach, but if taken with a meal, it would need to emerge rapidly as it and the food enter the small intestine. Otherwise, gluten would make mischief before the peptidase could kick into action. However, a pill taken before a meal wouldn’t remain in the intestine long enough to work.
Once in the right place at the right time, the enzyme would still need to penetrate the chemically complex mass of food being digested and find its specific molecular targets, Mowat says.
Even a peptidase that can knock out the 33–amino acid peptide might not fully preempt the immune response that defines celiac disease. Other amino acid sequences on other peptides also appear to trigger the pathological cascade, says Koning.
In the June 2002 Gastroenterology, he, Willemijn Vader, and their Leiden University colleagues identified several amino acid sequences on peptides that survive digestion and appear to trigger celiac disease in some children. In the September 2002 issue of that journal, Sollid and his colleagues described other amino acid sequences that act similarly in some adults. Exclusively targeting the 33–amino acid peptide, therefore, “is an oversimplification of the problem,” Koning and Vader contended in the Jan. 24 Science.
A separate problem applies to the strategy of inhibiting tTGase. This enzyme plays an important role in the normal repair of the stomach lining, so inhibiting it might reduce the body’s upkeep of that tissue and thereby cause intestinal bleeding, says Koning.
The safest possible therapy for celiac disease would probably be to block DQ2 and DQ8. Other immune molecules back up their functions in the body, Koning says.
Sollid, who helped identify the central role of DQ2 in celiac disease, agrees. Teams of researchers led by Khosla and Koning are currently focused on blocking DQ2.
However, Koning says, finding an effective inhibitor for DQ2 may take time because its binding site doesn’t grab onto molecules as tightly as the tTGase binding site does. Given how few of the disease-triggering immune molecules it takes to cause trouble, a drug might need to bind to DQ2 1,000 times as strongly as the gluten peptide does, Koning says.
Long pipeline
With such hurdles ahead, Sollid and other researchers estimate that it will take at least 5 years, and probably about 10, to bring to market any new drug for celiac disease.
Given that timeframe, researchers shouldn’t limit their current efforts to novel drugs, says John H. Griffin, chief scientific officer of Pharmix Corp., a small biotech firm in Redwood Shores, Calif. Numerous therapies that are already approved for other immune disorders of the gut–for instance, ulcerative colitis and the inflammatory bowel condition called Crohn’s disease–might prove capable of serving double duty against celiac disease, he suggests.
Because drug development and safety trials are already complete for such drugs, they could be tested with “no delay between the idea and the experiment,” Griffin says.
Unfortunately, at least one such attempt bore no fruit. In recent, unpublished experiments, Christophe Cellier of the Georges Pompidou European Hospital in Paris and his colleagues gave an anti-inflammatory drug designed to treat Crohn’s disease to three people with celiac disease, who then abandoned their gluten-free diets. But Cellier quickly halted the trial when the volunteers developed symptoms of celiac disease, he told Science News.
Researchers did report in the Nov. 7, 2002 Nature that statins, typically used to combat heart disease, provided a benefit to some patients with the immune disorder multiple sclerosis. If statins achieve this effect by clogging the receptors of misbehaving immune molecules, they may also confer benefits in celiac disease, says Khosla.
Since celiac disease is a lifelong condition, any drug that can help people manage it could offer the company that provides it a steady stream of revenue, says Griffin. “Given the true size of the patient population . . . that’s a substantial market opportunity,” he observes.
Yet even the discovery of a therapy that alleviates apparent symptoms may not permit people with the disease to eat a normal diet, Griffin adds. Researchers would have to follow people for years to determine whether any treatment mitigates the long-term dangers as well as does the diet alone or combined with the drug.
Such obstacles aren’t enough to discourage Khosla. He’s motivated by his son’s struggle through a childhood burdened by constant vigilance, occasional slips, and painful consequences.
Says Khosla, “When it’s late at night and I have one more thing to do, that’s what keeps me going.”
****************
If you have a comment on this article that you would like considered for publication in Science News, send it to editors@sciencenews.org. Please include your name and location.
To subscribe to Science News (print), go to https://www.kable.com/pub/scnw/
subServices.asp.
To sign up for the free weekly e-LETTER from Science News, go to http://www.sciencenews.org/subscribe_form.asp.







