Injecting a TB vaccine into the blood, not the skin, boosts its effectiveness
The BCG vaccine is notoriously bad at preventing the most common form of tuberculosis
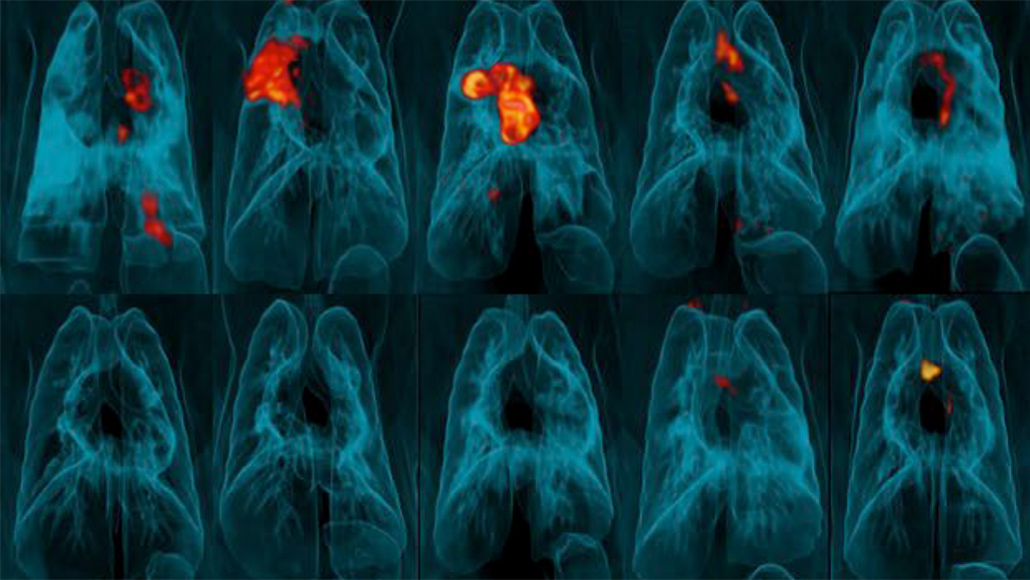
PET-CT scans of rhesus monkey lungs show spots of TB infection and tissue inflammation (red and orange). Monkeys that received a TB vaccine intravenously (bottom) were better protected than those who received it just under the skin (top).
University of Pittsburgh School of Medicine
Delivering a high dose of a vaccine against tuberculosis intravenously, instead of under the skin, greatly improves the drug’s ability to protect against the deadly disease, a new study finds.
Changing the typical dose and method of administration of the bacille Calmette-Guérin, or BCG, vaccine prevented TB in 90 percent of rhesus monkeys, researchers report online January 1 in Nature.
Most “astonishing” is that six of the 10 monkeys who received the IV vaccine never even developed an initial infection when exposed to TB, says Joel Ernst, an immunologist who specializes in TB at the University of California, San Francisco. Preventing infection, not just disease — called sterilizing immunity — is extremely rare with any TB vaccine, says Ernst, who was not involved in the study. Thwarting that infection means that no bacteria can reactivate to cause a latent or active TB infection.
The BCG vaccine has been around for nearly a century and is the only currently licensed TB vaccine. More than 150 countries, but not the United States, regularly use BCG to protect infants against some forms of TB. But the vaccine often fails to prevent the most common type of tuberculosis infection, in the lungs, in adolescents or adults.
Globally, TB infected 10 million people in 2018. It kills about 1.5 million a year, making it the most lethal infectious disease. Up to 13 million people in the United States have latent TB infection, which induces an immune response but hasn’t progressed to active tuberculosis. An experimental TB vaccine that could help protect people with the latent infection from developing active TB is in the works (SN: 9/25/18).
It’s been difficult to create an effective TB vaccine because the bacteria that cause the disease, Mycobacterium tuberculosis, enter cells, where they’re more protected from antibodies, which primarily attack outside cells. Fighting most intracellular infections requires immune cells called T cells to attack the infected cells, says immunologist Robert Seder of the National Institute of Allergy and Infectious Diseases Vaccine Research Center in Bethesda, Md.
Delivering the BCG vaccine just under the skin causes the body to make some T cells to fight TB. But not enough of these cells are created and get to where they need to be and stay there — the lungs, for example — limiting the vaccine’s effectiveness, says JoAnne Flynn, a microbiologist and immunologist at the University of Pittsburgh’s Center for Vaccine Research.
A malaria infection similarly requires T cells to fight the malaria parasite inside cells, Seder says. After his success with an intravenous malaria vaccine in another trial, researchers wondered: If they injected BCG vaccine directly into the blood, where it could travel throughout the body, would it trigger the creation of enough T cells in the tissues where the cells need to be?
Flynn, Seder and their colleagues tested five BCG formulations in macaques: a standard under-the-skin, or intradermal, human dose; a high dose given under the skin (100 times greater concentration than the human dose); an aerosol high dose administered with a mask; an intravenous high dose; and a combination of high-dose aerosol and standard-dose intradermal. Six months later, the research exposed the five differently vaccinated groups of macaques and a sixth unvaccinated control group to TB.
All of the unvaccinated, standard-dose intradermal and aerosol-vaccinated macaques developed the bacterial infection. The eight macaques that received the intradermal high dose did not have significantly better protection than those that got the standard dose, Flynn says. All but one of those eight developed infection, though two monkeys cleared it several weeks later. In contrast, six of 10 IV-vaccinated macaques never developed a TB infection, and three had fewer than 45 individual TB bacteria in the lungs, a very low amount, and went on to clear the infection.
One possible reason that the vaccine worked better when given intravenously is the high number of T cells induced by the IV vaccine — 100 times as many in those macaques’ airways compared with the intradermal and aerosol groups. Potentially more important is the discovery that the vaccine induced production of tissue-resident memory T cells, primed T cells in the tissue itself, not just the blood.
Punam Mangtani, an epidemiologist at the London School of Hygiene and Tropical Medicine, calls the research “a rare and exciting proof-of-concept study.”
Preventing TB in adolescents and adults is crucial, Flynn says, so the major question is whether this approach would be safe and effective in that target population. The only adverse effects seen in the macaques were a temporary, modest increase in inflammation. Ernst says one safety concern is whether intravenous BCG could induce a harmful inflammatory response in people with latent TB infection — about a quarter of the planet’s population. It’s not clear if this vaccine could help or harm those with latent infections, which the researchers plan to test in monkeys. If it could cause harm, screening before vaccination would be necessary.
For now, the next step is to test how low a dose still offers protection, Flynn says. “This study really provides us hope that a truly effective vaccine against TB is on the horizon,” she says. “I’ve been in the field for 30 years, and I feel we are making progress in really starting to understand the disease and vaccines that can prevent infection.”







