Things Just Mesh
Making stents even better at keeping arteries open
Fatty deposits on the insides of blood vessels pile up less obviously than in a bulging belly or widening hips, yet in some ways they carry as much weight. These tiny products of the process called atherosclerosis can block blood flow and oxygen to the heart, the brain, or other parts of the body.

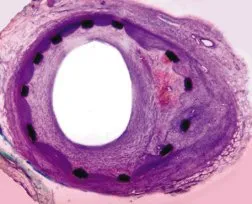
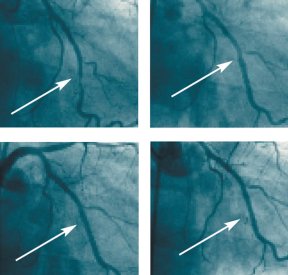
After using a variety of techniques to clear blocked arteries, physicians now commonly insert metal-mesh tubes called stents into arteries to hold them open and prevent loose bits of the fatty plaque from closing the vessel again or getting swept into the bloodstream, where they can obstruct other blood vessels.
Cardiologists credit stents with improving the safety of artery-clearing procedures and boosting the percentage of blood vessels that stay open afterward. Because of this success, physicians are now investigating whether stents can be used more widely. For example, the tubes might be inserted into blood vessels narrower than those treated today and into people so sick that they now receive major surgery to replace and bypass blocked vessels. What’s more, stents may find a role in blood vessels other than those servicing the heart. With each potential application of stents, however, the risks and benefits must be reassessed.
Problems identified in current applications of the devices provide several obstacles to broadening their use. Some stents trigger immune inflammation, and up to a third of them reclog–a process called restenosis–within a few months after their insertion. Restenosis is typically marked by chest pain, as blood flow is impaired and the heart protests a lack of oxygen. Doctors treat restenosis either by performing additional artery-clearing procedures or by open-heart surgery to bypass the blocked vessel. Such repeat operations are expensive, and recovery from them can be difficult for patients who are already ill.
“If we had the promise of a stent with a low risk of restenosis, we could treat difficult atherosclerotic lesions more confidently and probably send fewer people to bypass surgery,” notes Alan Heldman of Johns Hopkins University School of Medicine in Baltimore.
To develop a more long-lasting treatment, scientists are coating existing stents with polymers or gels containing drugs that block cell growth. “This is an exciting time for the whole field,” Heldman says.
Preventing heart disease
Doctors attack atherosclerosis to prevent it from triggering heart disease, one of the leading causes of death in the United States. Which treatment they choose depends on the severity of the artery clogging, often detected with angiograms, which are x-ray images of blood vessels. People with mild atherosclerosis may receive only medications designed to prevent blood clots and cholesterol-lowering drugs to block any further fatty buildup inside arteries. In people who have severe blockages in vessels serving the heart, cardiologists typically perform surgery to bypass the diseased arteries.
Since the early 1980s, physicians have used an intermediate procedure to treat people with moderate atherosclerosis. In this process, called angioplasty, a surgeon threads a tiny balloon inside an artery and inflates it, flattening the fatty deposits that narrow the artery. In the 1990s, doctors doing angioplasty started implanting a stent to improve the success of the procedure. For example, after Vice President Richard Cheney experienced chest pain in November 2000, he received a stent during angioplasty. Today, between 70 and 90 percent of people undergoing angioplasties get stents.
Recent studies have shown that the success rate for angioplasty is rising, even though physicians have been treating sicker and older patients than ever before.
According to researchers, most of that success is due to the widespread use of stents (SN: 2/3/01, p. 72: Success clearing clogged arteries).
In response to such findings, doctors are starting to treat severe atherosclerosis with angioplasty and a stent instead of bypass surgery. In the April 12 New England Journal of Medicine, an international team of researchers reported that each technique offers about the same degree of protection against death, stroke, and heart attack among people eligible for either procedure.
Angioplasty and a stent provided the less expensive approach but were more likely than bypass surgery to eventually lead to additional angioplasty or a follow-up round of bypass surgery, says study leader Patrick W. Serruys of the Academisch Ziekenhuis Rotterdam Dijkzigt in Rotterdam, the Netherlands. If restenosis in stents were less common, angioplasty with stents might become the preferred treatment for many people otherwise slated for open-heart surgery, he says.
“Some things get to be trendy–used widely just because they are new–but I don’t think that’s the case with stents,” says David O. Williams of Brown University in Providence, R.I. “The more we look, the better [stents’ performance] gets.”
The current problems arise because a newly implanted stent doesn’t stay bare for long. Eventually, the metal surface is covered by a thin layer of epithelial cells, which are the normal lining of blood vessels. The stent is thereby incorporated into artery walls, resembling steel bars embedded in concrete.
If healing stops there, restenosis is unlikely. However, sometimes the process goes into overdrive, causing smooth muscle cells from inside the blood-vessel wall to multiply and pile up inside the stent.
There are other causes of restenosis. The original problem of atherosclerosis can return, with fatty plaques building up inside the stent. Also, the stent can irritate the blood vessel, prompting white blood cells to swarm to the site. This can exacerbate atherosclerosis.
In large blood vessels with short blockages, less than 10 percent of stents clog. In small vessels, that rate can be more than 30 percent. Not all clogs in stents are large enough to dangerously block blood flow. Overall, about 12 to 15 percent of people who undergo angioplasty including a stent will need a follow-up procedure because of severe blockage. Because the stents are hard to spot with common imaging techniques, putting in another stent or performing bypass surgery can be tricky.
Drug coats
For these reasons, scientists and medical-device companies would like to develop stents resistant to restenosis. One approach is to coat stents with drugs. So far, drug-coated stents “look more promising than anything else we’ve seen,” says Peter Fitzgerald, a cardiologist at Stanford University.
In early September at the European Society of Cardiology Annual Congress in Stockholm, an international team of researchers reported tests of a stent coated with a polymer that releases sirolimus, an immune-suppressing drug that slows cell growth. The sirolimus coating boosted angioplasty success rates. After about 7 months, only 4 of the 120 patients who received a drug-coated stent had died, suffered a heart attack, developed blood clots, or needed a follow-up heart procedure. After the same amount of time, 32 of the 118 people with a bare-metal stent had experienced one of those adverse events. Researchers didn’t spot restenosis in angiograms of any patient implanted with the drug-coated stent, which is made by Cordis, a Johnson and Johnson company in Miami Lakes, Fla.
Cook, a company headquartered in Bloomington, Ind., is testing metal stents coated directly with the anticancer drug paclitaxel. Like most anticancer drugs, paclitaxel kills dividing cells, a process that in this case might prevent smooth muscle cells within an artery from overgrowing a stent.
In a study of 177 people receiving either bare-metal or paclitaxel-coated stents, 27 percent of the bare stents narrowed by more than half within 6 months. In contrast, 12 percent of stents coated with a low dose of the drug and just 4 percent of stents with high-dose paclitaxel were blocked to this degree.
The company reports that the buildup inside the stents was least in the high-dose paclitaxel stents, slightly more in the low-dose stents, and most pronounced in the bare stents. The company presented these results in Washington, D.C., at a September meeting on transcatheter cardiovascular therapeutics.
This month at the annual meeting of the American Heart Association in Anaheim, Calif., researchers working with the Cook stent reported results from a trial of 192 European patients. In this study, 21 percent of the bare-metal stents narrowed by more than half after surgery. The researchers tested several doses of paclitaxel released from the stents. The more paclitaxel was released, the more benefit.
Just 3.1 percent of the stents releasing the highest dose of paclitaxel narrowed by half within 6 months. There was no reported side effect from the drug. The average percentage of restenosis in each stent after 6 months was 34 percent in the group treated with bare-metal stents and 14 percent in those people implanted with the high-dose paclitaxel stent.
In another study released at the September meeting, Ivan De Scheerder of University Hospitals in Leuven, Belgium, reported success in placing paclitaxel-coated stents inside already implanted, bare-metal stents that had repeatedly reclogged. Six months after doctors implanted new stents inside the old ones in 21 patients, there was no sign of restenosis in any patient.
In a less formal setting, Boston Scientific Corp., a Boston-based company that makes stents, recently reported to stock analysts promising results from an initial trial of a stent coated with a paclitaxel-containing polymer. Larger studies of these paclitaxel-releasing stents are now under way.
These “are significant benefits,” says Fitzgerald. “There’s not a lot in our armament that’s gotten people this excited. I think we’ll adopt coated stents like crazy.” Still, there may yet be unexpected roadblocks, Fitzgerald and other researchers caution.
Premature celebration?
While cardiologists’ celebration over the early results with coated stents is understandable, says Renu Virmani of the Armed Forces Institute of Pathology in Washington, D.C., “this is a premature celebration. We need to wait for the long term, perhaps 2 or 3 years.”
Virmani bases her caution in part on history. In the late 1990s, researchers showed that stents impregnated with radioactive elements are effective in reducing the buildup of new cells inside the stent. Over the past year or so, however, several research teams have shown limits to the proposed benefits.
The radiation kills cells within the stent, preventing restenosis, but it also damages tissue upstream and downstream of the mesh tube, researchers have discovered. In the process of healing that damage, cells in these areas grow rapidly, narrowing the blood vessels at either end of the stent in what scientists have called a “candy-wrapper” effect. So far, there is no sign of such an effect around drug-coated stents.
Virmani says that her first reservation about these devices is that “once the drug disappears, there will be no benefit.” Animal trials of the sirolimus-coated stent suggest that the drug lasts for 15 or 45 days, depending on the formulation.
Virmani’s also concerned that the drug coatings will slow the incorporation of the stent within the vessel’s walls. The thin covering of epithelial cells reduces the risk that a metal stent will trigger blood clotting, she says.
This potential effect on blood clotting “is a theoretical concern, not yet seen,” says Fitzgerald. However, he agrees that researchers should carefully track the incidence of blood clots.
“Restenosis rarely kills anybody. They walk into the hospital and say, ‘Doc, my chest is hurting,'” Fitzgerald says. Their restenosis can then be treated.
On the other hand, blood clots can break off, clog an artery, and cause a heart attack or stroke without warning. Fitzgerald says, “We really have to look carefully at these studies and make sure what we’re so excited about holds up over the long term.”
Next-generation devices
Prompted in part by the excitement over the success of drug-coated stents, research into next-generation devices is moving ahead quickly. Those are being made with new coatings or new materials.
In one approach, researchers are changing the way that stents release a drug. Right now, chemicals associated with the stent start entering the blood as soon as doctors put the device into the body. But overactive growth of smooth muscle cells and inflammation are most likely to start a few days later. If a drug instead could remain bound for a day or two after implantation, restenosis might be more effectively deterred, Fitzgerald says.
Given the need for the stent to become incorporated within the blood vessel, other researchers are trying to find materials that will block the growth of smooth muscle cells but let epithelial cells grow normally.
Robert J. Levy of the Children’s Hospital of Philadelphia and his colleagues are developing stents with a coating containing genes that, if absorbed into nearby cells, might slow atherosclerosis. “Because the stents are implanted at areas of major disease activity, it’s an opportunity to treat local disease beyond the mechanical benefit of keeping the artery open,” he says.
Last November, Levy and his colleagues reported that they could use stents to deliver marker genes to cells in blood vessels (SN: 11/18/00, p. 325). These genes had no particular function but were easy to detect in cells that took them up, he explains. The researchers haven’t yet decided which genes might provide effective therapy.
The stent coating that Levy used in his initial studies triggered some inflammation. At the American Heart Association meeting this month, Levy reported tests that used a stent coating of collagen, the rubbery tissue of connective tissue and bones. It delivered genes to the blood vessel immediately around the stent while inducing little inflammation.
Stents themselves are the targets for improvements. Since the chance of restenosis is highest in the first several months after surgery, researchers have tried to develop stents that will degrade after the blood vessel has healed from angioplasty. Getting materials that do this has been surprisingly difficult, however, because many of the biodegradable stents have triggered immune responses and inflammation when tested in animals.
A new biodegradable stent seems to provoke less of an immune response. In the July 25, 2000 Circulation, Hideo Tamai of the Shiga Medical Center for Adults in Moriyama, Japan, and his colleagues reported that a stent made from a biodegradable polymer was safe and effective in patients who were undergoing angioplasty.
At the American College of Cardiology meeting in Los Angeles last February, the team reported that 1 year after receiving a biodegradable stent, 7 of 36 patients showed signs of restenosis, about the same as in patients getting a traditional metal stent. The scientists plan to continue observing these patients to see how the new stents degrade and what the long-term consequences are.
A stent that will degrade over time is less likely than a metal tube to interfere with future angioplasty or bypass surgery, says Antonio Colombo of the Centro Cuore Columbus in Milan. With Tamai’s work, he says, “a number of former problems have been resolved and a new approach to coronary stenting is emerging.”
Biodegradable stents could also serve as a platform for drug or gene delivery.
Overall, cardiologists are hopeful that drug-coated stents–and their successors–will address the serious problem of atherosclerosis even more safely and effectively than angioplasty has to date. “Drug-coated stents work, and our patients will see benefits,” says Heldman. With cardiologists considering broad expansions of stents’ widespread use today, he says, preventing restenosis is more important than ever.







