Trilayer graphene exhibits quantum effect
Three could be the magic number for making spintronic devices from thin carbon sheets
When stacked like lasagna noodles, the thinnest material on the planet changes what it can do — and what those trying to make devices out of it might do with it.
Electrons act funny in three sheets of atom-thick carbon, or graphene, arranged just so. The particles’ masses seem to change in an unusual way that depends on the electrons’ speed.
“The slower these particles move, the heavier they become,” says Igor Zaliznyak, a physicist at Brookhaven National Laboratory in Upton, N.Y., whose team reports this property online September 25 in Nature Physics.
That’s the opposite of electrons in metal, which, in accordance with Einstein’s theory of relativity, gain mass when moving faster.
But strange subatomic behavior is par for the course in graphene, a flat wonderland of quantum weirdness (SN: 8/13/11, p. 26). In a single sheet of graphene, electrons race around as if they have no mass at all. Add a second layer of graphene, and electrons once again behave as if they have mass. Zaliznyak’s experiment confirms predictions of how electrons should behave when a third layer is slapped on top.
Graphene’s bizarre physics starts with its electrons’ penchant for communal living. Confined within the structure of this material, graphene’s electrons come together and form a single quantum entity. In 2005, scientists revealed this collective quantum state by placing a slice of graphene in a strong magnetic field. As the field was ramped up, the resistance of the material increased in sudden, discrete jumps — a sign of electrons acting in concert known as the quantum Hall effect.
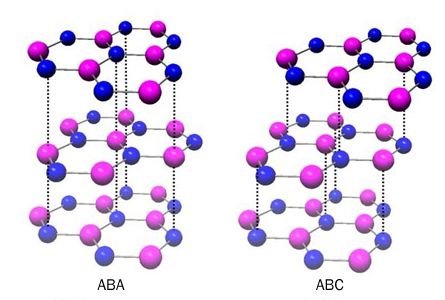
In 2006 the quantum Hall effect, common in semiconductors, was found in two layers of graphene joined together. But it’s been harder to spot in trilayer graphene — partly because three sheets of this carbon chicken wire can be layered together in more than one way, says Walter Escoffier, a physicist at the CNRS Laboratoire National des Champs Magnétiques Intenses in Toulouse, France.
“This effect was predicted for trilayer graphene but never experimentally observed until now,” he says. Escoffier is a member of the team that reported the effect in the Sept. 16 Physical Review Letters.
Finding the right arrangement of layers is a game of molecular Tetris. Line up the top and bottom sheets of honeycombed atoms, sandwich-style, and the material behaves like a metal. Only when layers step upward at a slant, like stairs, does the quantum weirdness begin.
“By shifting one of the layers a little bit, you change its properties very dramatically,” says Jeanie Lau, a physicist at the University of California, Riverside.
Trilayer graphene’s unusual electrons may be useful for spintronic devices, which exploit not the charge of an electron but a quantum property called spin. Lining up the spins of electrons in three layers of graphene should be easier than in a single sheet, says Zaliznyak.
Adding even more layers to graphene could reveal new and potentially useful quantum curiosities, says Lau. Her team is playing with four layers of carbon and thinking about adding even more. At some point, graphene stacked high enough should start to again behave like its more mundane cousin, graphite. But the line between funky quantum lasagna and ordinary pencil lead hasn’t yet been drawn.