Year in review: Memories vulnerable to manipulation
Experiments reveal how brain forms, stores recollections
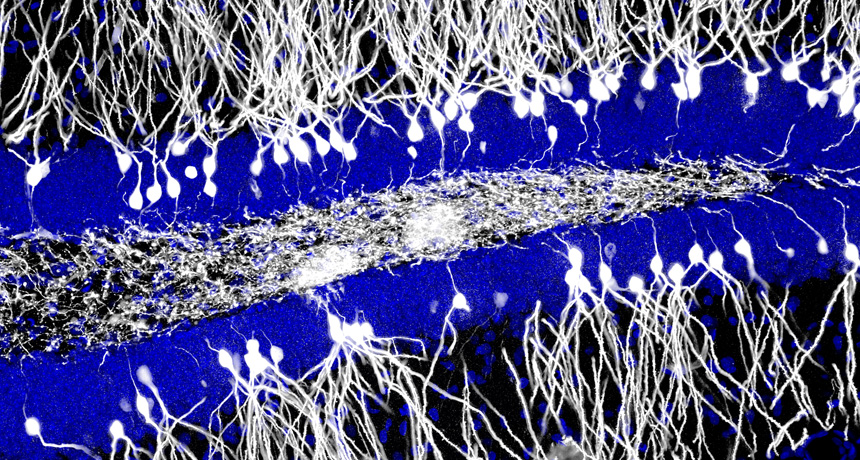
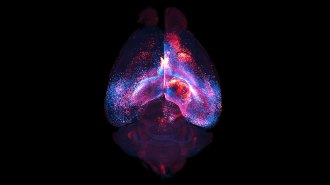
JUMBLED The hippocampus (blue, mouse brain shown) churns out neurons (white) early in life that may disrupt memories from infancy.
Courtesy of Jason Snyder
5
Memories can often seem hazy, but the brain creates and destroys them with exacting precision. New experimental results reported in 2014 helped bring scientists closer to understanding how the brain manipulates memories to make sense of
the world.
The results have practical implications, too: By picking apart the memory process, scientists may ultimately be able to intervene in cases when bad memories become problematic. “Right now it may sound like science fiction,” says neuroscientist Susumu Tonegawa of MIT. But in the next few decades, scientists and clinicians may develop sophisticated ways of tapping into certain brain regions to ease bad memories that can contribute to problems such as post-traumatic stress disorder or depression, he says.
This year, Tonegawa and his colleagues transformed bad mouse memories into good ones (SN: 10/4/14, p. 6). Relying on a technique called optogenetics, the researchers selectively tagged the nerve cells that stored a bad memory of foot shocks. Those tags allowed the researchers to evoke the bad memory at any time by shining a laser light onto the cells via implanted optical fibers.
By calling up the bad memory while male mice were in a pleasant situation (in the enjoyable presence of females), the researchers took some of the sting out of the formerly traumatic memory. Similarly, the researchers sullied a good memory by calling it up as the mice were shocked.
The cells manipulated in this memory switch were in a particular spot of the hippocampus, a brain structure involved in all sorts of memories. One notable example comes from the hippocampus’s “place cells,” which recollect precise physical locations. The discovery of those cells won John O’Keefe of University College London a share of this year’s Nobel Prize in physiology or medicine (SN: 11/1/14, p. 15).
While certain kinds of hippocampus cells are important for remembering, others may be important for forgetting, a provocative study of mice found (SN: 6/14/14, p. 7). Unlike many other regions of the brain, the hippocampus continually churns out newborn nerve cells, or neurons, particularly during early life. But just how these newborn cells contribute to memory hasn’t been clear. The new study suggests that early on, the flood of newborn cells might destabilize memories, which could explain why humans, rats and other animals don’t remember much from their infancy.
Mouse pups that had been treated to produce fewer neurons better remembered a room where they received shocks than did pups that churned out the normal number of neurons, Sheena Josselyn of the Hospital for Sick Children in Toronto and her colleagues found. Cranking up the neuron birthrate made adult mice more forgetful. In contrast, young guinea pigs and degus, rodents whose brains don’t churn out new neurons early in life, didn’t show signs of amnesia.
Newborn neurons may elbow out the preexisting infrastructure that held memories in place, causing the animal to forget. When the birthrate of these neurons slows down, the brain’s memory machinery may become more stable, capable of forming long-lasting memories.
Most memory manipulations have occurred in lab animals, which can be genetically engineered and given drugs to change brain cell behavior. A counterexample comes from a small study on depressed humans who received electroconvulsive therapy: The electric jolt weakened memories that had been called to mind just before treatment, a team led by Marijn Kroes of Radboud University Nijmegen in the Netherlands found (SN: 2/8/14, p. 10).
Electroconvulsive therapy is unlikely to become a treatment for people suffering from intrusive negative memories. Nor do clinicians have the ability to specifically manipulate neurons that store a memory or selectively dial up or down neuron birth in people. But scientists are working on new techniques that might ultimately allow clinicians to tap into these memory processes and fix them when they go awry, Tonegawa says.