With only rare exceptions, every organism constructs proteins from just 20 building blocks called amino acids. Recently, however, researchers modified Escherichia coli bacteria so that the single-celled organisms also make an alien amino acid and incorporate it into proteins (SN: 1/25/03, p. 53: Available to subscribers at Unnatural Biochemistry: Bacteria make and use an alien amino acid).
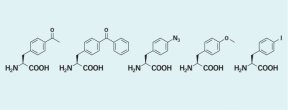
Now, the same scientists have used genetic engineering techniques to coerce more-complicated organisms into placing unnatural amino acids into proteins. Led by Peter G. Schultz of the Scripps Research Institute in La Jolla, Calif., the team created yeast cells that add one of five unnatural amino acids to their natural 20-piece construction set. To give the yeast this capability, the researchers altered the cells’ biochemical machinery that identifies amino acids and puts them into particular locations within a protein.
Unlike the E. coli on which the team previously reported, the yeast doesn’t manufacture synthetic amino acids itself. Instead, it takes the molecules up from the nutrient mixture in which it’s grown. The researchers describe this work on Saccharomyces cerevisiae in the Aug. 15 Science.
The Scripps team suggests that scientists could use this technology to produce proteins with new or enhanced properties. For example, improved drugs might result. The development may also improve the study of protein function in yeast, the scientists report.
The unnatural amino acids used by the engineered yeast have various useful properties, says Scripps team member T. Ashton Cropp. For example, when activated by light, two of these amino acids bond proteins together, a function that could prove valuable in studies of protein interactions. Another of the novel amino acids might help researchers using X-ray crystallography studies to elucidate protein structure.
“It is another brilliant achievement from the Schultz group,” says Jack F. Kirsch of the University of California, Berkeley. “The potential applications are endless.”
“This technology will be very powerful and very useful,” agrees Hung-wen (Ben) Liu of the University of Texas at Austin. He says he hopes the Scripps team will make the technology available to other scientists for basic biological investigations.
The method should also work on organisms even more complex than yeast, says the study’s lead author, Jason W. Chin, who is now at the Medical Research Council Laboratory of Molecular Biology in Cambridge, England. That’s because yeast contain protein-building machinery that’s similar to that in multicellular organisms, from peas to people. Yeast cells and those of the more complex organisms are classified as eukaryotes because, unlike bacteria, they contain distinct nuclei and organelles.
In fact, says Cropp, the team has already begun experiments in which it’s trying to get human cells to use unnatural amino acids when making proteins.
****************
If you have a comment on this article that you would like considered for publication in Science News, send it to editors@sciencenews.org. Please include your name and location.







