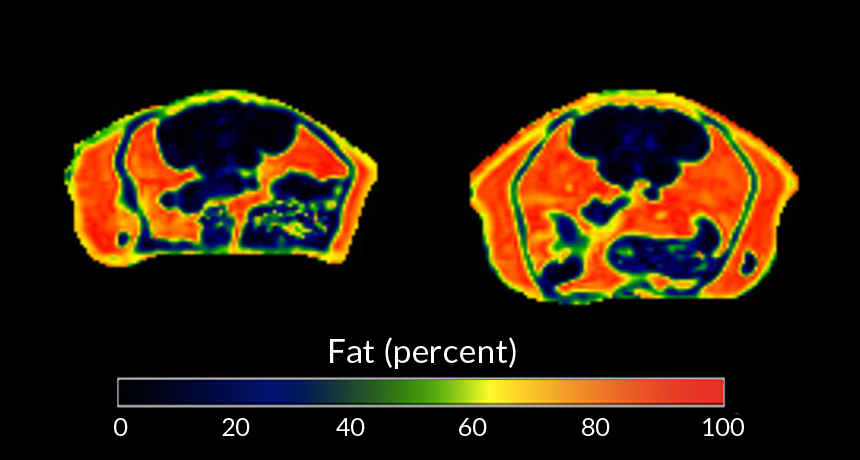
PLUMP UP Mice treated as babies with low doses of penicillin (right, MRI image of an animal’s abdomen shown) nearly doubled their body fat when fed high-fat diets as adults. Mice that didn’t get antibiotics as babies (left) didn’t pack on as much fat on the same diet.
L.M. Cox and Sungheon G. Kim
Antibiotics given to infants may have life-long consequences, a study of mice suggests.
Low doses of antibiotics given to pregnant mice and to their newborns led baby mice to become obese as adults, researchers report August 14 in Cell. The effect was not due to the drugs themselves but to the disruption of the rodents’ gut microbiome, the community of microbes living in the mice’s intestines.
“We’re using antibiotics as if there were no cost,” says microbiologist Martin Blaser of New York University, who led the study. “The costs are not immediate but may be long-term.”
Blaser’s team has previously found that antibiotics alter the microbiome and that those changes can lead to inflammation and promote weight gain in animals. In the new study, the researchers wanted to find out what happens if a baby takes antibiotics when the microbiome is establishing itself. The microbiome is known to help train the immune system.
Microbiologist Laura Cox in Blaser’s lab and colleagues treated pregnant mice with low doses of penicillin, continuing the treatment once the pups were born until the baby mice weaned, at about 4 weeks old. That is equivalent to about the first year of a human baby’s life. Other mice slurped the drug in their drinking water until they were 8 weeks old, while others were on penicillin for life or never were exposed to the antibiotic.
Gut bacteria compositions in antibiotic-treated mice were different from those of mice that didn’t get the drug. When the drug was halted at weaning, the pups’ microbes became normal by the time they were about 8 weeks old.
But the drug use did have some lasting effects. Animals that got antibiotics early in life had changes in their immune system and metabolism that lasted into adulthood. Antibiotic-treated baby mice grew up to become plump adults. And when adults ate a high-fat diet, those that got penicillin as babies became obese: Female mice nearly doubled their fat mass.
Those animals also had elevated levels of insulin in their blood, a warning sign of diabetes. The results indicate that early exposure to antibiotics can change the development of the body’s metabolic systems, says Cox.
To determine whether the alterations were due to the side effects of the drugs or the temporary changes in the microbiome, the researchers transplanted bacteria from baby mice into the intestines of mice raised without any bacteria.
When the bacteria came from life-long antibiotic-treated mice, the transplant recipients got fat. Mice stayed slim if they got microbiome transplants from animals that hadn’t been treated with antibiotics. Those results suggest that the altered microbe mix was responsible for the weight gain.
The researchers don’t yet know whether shorter courses of antibiotics, like those used to treat babies’ ear infections, will have the same effect, Cox says.
Epidemiologist Anita Kozyrskyj of the University of Alberta in Edmonton, Canada, thinks early antibiotic exposure may affect human children similarly. She and colleagues report August 5 in the International Journal of Obesity that boys who got short courses of antibiotics before they were a year old weighed more as middle schoolers than kids who didn’t get antibiotics early on. Girls who took antibiotics as infants also tended to be heavier, but other factors, such as whether their mothers were overweight during pregnancy, seem more important in influencing girls’ weight.
The data are compelling that early-life experiences with antibiotics can have long-term consequences, says Patrick Seed, a pediatrician and molecular microbiologist at Duke University. He worries that parents may refuse life-saving antibiotics for their children when they learn that the drugs might predispose kids to obesity. “We have to have restraint with antibiotics, but we can’t completely fear them,” he says.
Besides, not every baby who takes penicillin will grow up to be fat. “We know that antibiotics are not a universal equation for obesity,” Seed says. Diet and other lifestyle factors are important, too.
Seed says that learning more about how microbes influence human development might help researchers devise ways to treat serious infections without damaging a child’s future health.







