POWERING THE FUTURE New chemistries and designs promise to take batteries into the 21st century to store energy from solar and wind farms and send energy to homes and businesses.
Vanatchanan/Shutterstock
Everybody wants more juice from their batteries. Smartphones and laptops always need recharging. Electric car drivers must carefully plan their routes to avoid being stranded far from a charging station. Anyone who struggles with a tangle of chargers every night would prefer a battery that can last for weeks or months.
For researchers who specialize in batteries, though, the drive for a better battery is less about the luxury of an always-charged iPad (though that would be nice) and more about kicking our fossil fuel habit. Given the right battery, smog-belching cars and trucks could be replaced with vehicles that run whisper-quiet on electricity alone. No gasoline engine, no emissions. Even airplanes could go electric. And the power grid could be modernized to use cheaper, greener fuels such as sunlight or wind even on days when the sun doesn’t shine bright enough or the wind doesn’t blow hard enough to meet electricity demand.
A better battery has the potential to jolt people into the future, just like the lithium-ion battery did. When they became popular in the early 1990s, lithium-ion batteries offered twice as much energy as the next best alternative. They changed the way people communicate.
“What the lithium-ion battery did to personal electronics was transformational,” says materials scientist George Crabtree, director of the Joint Center for Energy Storage Research at Argonne National Laboratory in Illinois. “The cell phone not only made landlines obsolete for many, but [the lithium-ion battery] put cameras and the internet into the hands of millions.” That huge leap didn’t happen overnight. “It was the sum of many incremental steps forward, and decades of work,” says Crabtree, who coordinates battery research in dozens of U.S. labs.
Lithium-ion batteries have their limits, however, especially for use in the power grid and in electric vehicles. Fortunately, like their Energizer mascot, battery researchers never rest. Over the last 10 years, universities, tech companies and car manufacturers have explored hundreds of new battery technologies, reaching for an elusive and technically difficult goal: next-generation batteries that hold more energy, last longer and are cheaper, safer and easier to recharge.
A decade of incremental steps are beginning to pay off. In late 2017, scientists will introduce a handful of prototype batteries to be developed by manufacturers for potential commercialization. Some contain new ingredients — sulfur and magnesium — that help store energy more efficiently, delivering power for longer periods. Others will employ new designs.
“These prototypes are proof-of-principle batteries, miniature working versions,” Crabtree says. Getting the batteries into consumer hands will take five to 10 years. Making leaps in battery technology, he says, is surprisingly hard to do.
Power struggle
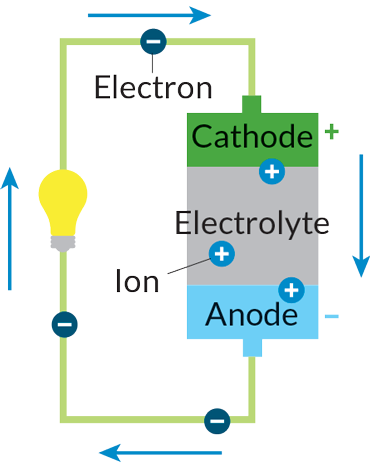
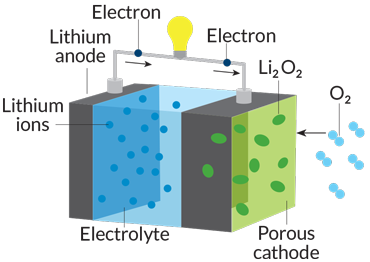
Batteries operate like small chemical plants. Technically, a battery is a combination of two or more “electrochemical cells” in which energy released by chemical reactions produces a flow of electrons. The greater the energy produced by the chemical reactions, the greater the electron flow. Those electrons provide a current to whatever the battery is powering — kitchen clock, smoke alarm, car engine.
To power any such device, the electrons must flow through a circuit connecting two electrodes, known as an anode and a cathode, separated by a substance called an electrolyte. At the anode, chemical oxidation reactions release electrons. At the cathode, electrons are taken up in reduction reactions. The electrolyte enables ions created by the oxidation and reduction reactions to pass back and forth between the two electrodes, completing the circuit.
Depending on the materials used for the electrodes and the electrolyte, a battery may be recharged by supplying current that drives the chemical reactions in reverse. In creating new recipes for a rechargeable electrochemical soup, though, battery researchers must beware of side reactions that can spoil everything.
“There’s the chemical reaction you want — the one that stores energy and releases it,” Crabtree says. “But there are dozens of other … reactions that also take place.” Those side reactions can disable a battery, or worse, lead to a risk of catastrophic discharge. (Consider the recent fires in Samsung’s Galaxy Note 7 smartphones.)
Early versions of the lithium-ion battery from the 1970s carried an anode made of pure lithium metal. Through repeated use, lithium ions were stripped off and replated onto the anode, creating fingerlike extensions that reached across to the cathode, shorting out the battery. Today’s lithium-ion batteries have an anode made of graphite (a form of carbon) so that loose lithium ions can snuggle in between sheets of carbon atoms.
Lithium-ion batteries were originally developed with small electronics in mind; they weren’t designed for storing electricity on the grid or powering electric vehicles. Electric cars need lots of power, a quick burst of energy to move from stop to start. Electric car manufacturers now bundle thousands of such batteries together to provide power for up to 200 miles before recharging, but that range still falls far short of what a tank of gas can offer. And lithium-ion batteries drain too quickly to feed long hours of demand on the grid.
Simply popping more batteries into a car or the grid isn’t the answer, Crabtree says. Stockpiling doesn’t improve the charging time or the lifetime of the battery. It’s also bulky. Carmakers have to leave drivers room for their passengers plus some trunk space. To make electric vehicles competitive with, or better than, vehicles run by internal-combustion engines, manufacturers will need low-cost, high-energy batteries that last up to 15 years. Likewise, grid batteries need to store energy for later use at low cost, and stand up to decades of use.
“There’s no one battery that’s going to meet all our needs,” says MIT materials scientist Yet-Ming Chiang. Batteries needed for portable devices are very different from those needed for transportation or grid-scale storage. Expect to see a variety of new battery types, each designed for a specific application.
Switching to sulfur
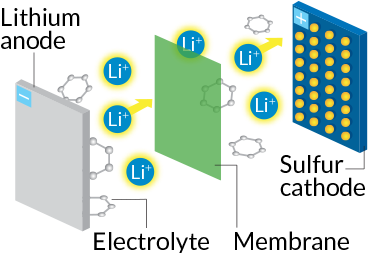
For electric vehicles, lithium-sulfur batteries are the next great hope. The cathode is made mainly of sulfur, an industrial waste product that is cheap, abundant and environmentally friendly. The anode is made of lithium metal.
During discharge, lithium ions break away from the anode and swim through the liquid electrolyte to reach the sulfur cathode. There, the ions form a covalent bond with the sulfur atoms. Each sulfur atom bonds to two lithium ions, rather than just one, doubling the number of bonds in the cathode of the battery. More chemical bonds means more stored energy, so a lithium-sulfur battery creates more juice than a lithium-ion one. That, combined with sulfur’s light weight, means that, in principle, manufacturers can pack more punch for a given amount of weight, storing four or five times as much energy per gram.
Ultimately, that upgrade could boost an electric vehicle’s range up to as much as 500 miles on a single charge. But first, researchers have to get past the short lifetime of existing lithium-sulfur batteries, which, Crabtree says, is due to a loss of lithium and sulfur in each charge-discharge cycle.
When lithium combines with sulfur, it also forms compounds called polysulfides, which quickly gum up the battery’s insides. Polysulfides form within the cathode during battery discharge, when stored energy is released. Once they dissolve in the battery’s liquid electrolyte, the polysulfides shuttle to the anode and react with it, forming a film that renders the battery useless within a few dozen cycles — or one to two months of use.
At Sandia National Laboratories in Albuquerque, N.M., a team led by Kevin Zavadil is trying to block the formation of polysulfides in the electrolyte. The electrolyte consists of salt and a solvent, and current lithium-sulfur batteries require a large amount of electrolyte to achieve a moderate life span. Zavadil and his team are developing “lean” electrolyte mixtures less likely to dissolve the sulfur molecules that create polysulfides.
Described September 9 in ACS Energy Letters, the new electrolyte mix contains a higher-than-usual salt concentration and a “sparing” amount of solvent. The researchers also reduced the overall amount of electrolyte in the batteries. In test runs, the tweaks dropped the concentration of polysulfides by several orders of magnitude, Zavadil says.

“We [also] have some ideas on how to use membranes to protect the lithium surface to prevent polysulfides from happening in the first place,” Zavadil says. The goal is to produce a working prototype of the new battery — one that can last through thousands of cycles — by the end of 2017.
At the University of Texas at Austin, materials engineer Guihua Yu, along with colleagues at Zhejiang University of Technology in Hangzhou, China, is investigating another work-around for this battery type: replacing the solid sulfur cathode with an intricate structure that encapsulates the sulfur in an array of nanotubes. Reported in the November issue of Nano Letters, the nanotubes that encase the sulfur are fashioned from manganese dioxide, a material that can attract and hold on to polysulfides. The nanotubes are coated with polypyrrole, a conductive polymer that helps boost the flow of electrons.
This approach reduces buildup and boosts overall conductivity and efficiency, Yu says. So far, with the group’s new architecture, the battery loses less than 0.07 percent of its capacity per charge and discharge cycle. After 500 cycles, the battery maintained about 65 percent of its original capacity, a great improvement over the short lifetime of current lithium-sulfur batteries. Still, for use in electric vehicles, scientists want batteries that can last through thousands of cycles, or 10 to 15 years.
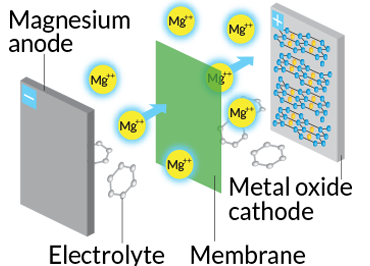
Scientists at Argonne are constructing another battery type: one that replaces lithium ions at the anode with magnesium. This switch could instantly boost the electrical energy released for the same volume, says Argonne materials engineer Brian Ingram, noting that a magnesium ion has a charge of +2, double that of lithium’s +1. Magnesium’s ability to produce twice the electrical current of lithium ions could allow for smaller, more energy-dense batteries, Ingram says.
Magnesium comes with its own challenge, however. Whereas lithium ions zip through a battery’s electrolyte, magnesium ions slowly trudge. A team of researchers at Argonne, Northwestern University and Oak Ridge National Laboratory shot high-energy X-rays at magnesium in various batteries and learned that the drag is due to interactions with molecules that the magnesium attracts within the electrolyte. Ingram and his group are experimenting with new materials to find a molecular recipe that reduces such drag.
Ingram’s team is trying to nudge its “highly functioning, long-lasting” prototype to 3 volts by December. Today’s typical lithium-ion battery has 3.8 to 4 volts. At 3 volts, Ingram says, the magnesium battery would pack more power than a 4-volt lithium-ion battery and “create a tremendous amount of excitement within the field.”
Going with the flow
Together, transportation and the electricity grid account for about two-thirds of U.S. energy use. But today, only 10 percent of the electricity on the grid is from renewable sources, according to the U.S. Energy Information Administration. If wind and solar power are ever to wrestle energy production away from fossil fuels, big changes must happen in energy storage.
What is needed, Crabtree says, is a battery that can store energy, and lots of it, for later use. “Though the sun shines in the middle of the afternoon, peak demand comes at sunset when people go home, turn on lights and cook dinner,” he says.
To reliably supply electricity at night or on cloudy, windless days requires a different type of battery. By design, flow batteries fit the bill. Instead of having solid electrodes, flow batteries store energy in two separate tanks filled with chemicals — one positively charged, the other negatively charged. Pumps move the liquids from the tanks into a central chamber, or “stack,” where dissolved molecules in the liquids undergo chemical reactions that store and give up energy. A membrane located in the stack keeps the positive and negative ions separated.
Flow batteries can store energy for a long time and provide power as needed. Because the energy-storing liquids are kept in external tanks, the batteries are unlikely to catch fire, and can be built large or small depending on need. To store more power, use a larger tank.
So far, however, flow batteries are expensive to make and maintain, and have had limited use for providing backup power on the grid. Today’s flow batteries are packed with rare and toxic metal components, usually vanadium. With many moving parts — tanks, pumps, seals and sensors — breakdowns and leakage are common.
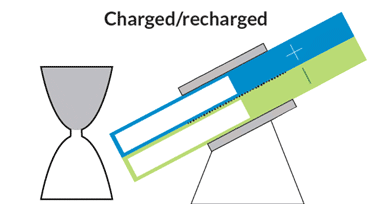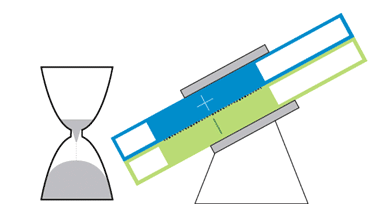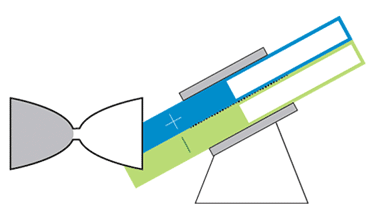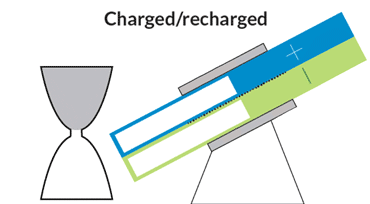
At MIT, Chiang and colleagues are developing flow batteries that can bypass those drawbacks. One, an hourglass flow battery, does away with the need for costly and troublesome pumps. The stack where the chemical reactions occur is in the constricted middle, with tanks at either end. Gravity allows the liquids to flow through the stack, like sand in an hourglass. A motor adjusts the battery’s angle to speed or slow the flow.
The hourglass design is like a “concept car,” Chiang says. Though the final product is likely to take a slightly different shape, the design could serve as a model for future flow batteries. Simply changing the tilt of the device could add a short infusion of power to the grid during periods of peak demand, or slowly release energy over a period of many hours to keep air conditioners and heaters running when the sun is down.
In another design, the group has replaced vanadium with sulfur, which is inexpensive and abundant. Dissolved in water (also cheap and plentiful), sulfur is circulated into and out of the battery’s stack, creating a reaction that stores or gives up energy, similar to commercial flow batteries. The group is now refining the battery, first described in 2014 in Nano Letters, aiming for higher levels of energy.
Another challenge in developing flow batteries is finding ways to keep active materials confined to their tanks. That’s the job of the battery membrane, but because the organic molecules under consideration for battery use are almost always small, they too easily slip through the membrane, reducing the battery’s lifetime and performance.
Rather than change the membrane, a group led by chemist Joaquín Rodríguez-López of the University of Illinois at Urbana-Champaign devised ways to bulk up the battery’s active materials by changing their size or configuration. The scientists linked tens to millions of active molecules together to create large, ringed structures, long strings of molecules hooked onto a polymer backbone, or suspensions of polymers containing up to a billion molecules, they reported in the Journal of the American Chemical Society in 2014.
With the oversized molecules, even “simple, inexpensive porous membranes are effective at preventing crossover,” Crabtree says. A prototype flow battery that provides low-cost power and lasts 20 to 30 years is expected to be completed in the coming year.
Getting air
Looking beyond 2017, scientists envision a new generation of batteries made of low-cost, or even no-cost, materials. The lithium-air battery, still in early development, uses oxygen sucked in from the atmosphere to drive the chemical reaction that produces electricity. In the process, oxygen combines with lithium ions to form a solid compound (lithium peroxide). During charging, solid oxygen reverts back to its gaseous form.
“Lithium-air potentially offers the highest energy density possible,” says MIT materials engineer Ju Li. “You basically react lithium metal with oxygen in air, and in principle you get as much useful energy as gasoline.”
Lithium-air has problems, though. The batteries are hard to recharge, losing much of their power during the process. And the chemical reaction that powers the battery generates heat, cutting the battery’s energy-storage capacity and life span.
Using electron microscopy to study the reaction products of a lithium-air prototype, Li and his group came up with a possible solution: Keep oxygen in a solid form sealed within the battery to prevent the oxygen from forming a gas. By encasing oxygen and lithium in tiny glasslike particles, the scientists created a fully sealed battery. The new strategy, published July 25 online in Nature Energy, curbed energy loss during recharging and prevented heat buildup.
“If it works on a large scale, we would have an electrical vehicle that’s competitive with gasoline-driven cars,” Li says. Reaching that goal would be a big step toward a greener planet.
This article appears in the January 21, 2017, issue of Science News with the headline, “Charging the future: Researchers look for ways to electrify the battery business.”
Editor’s Note: This story was modified on January 25, 2017, to clarify the distinction between power and energy.