Stealing a page from Star Trek, research teams in the United States and abroad are independently developing ultrasound devices that would enable caregivers to stop internal bleeding or excise tumors without a single incision. The benefits would cascade into fewer infections, faster healing, and saved lives.
“In the long run, it may be the way of choice because it’s an outpatient technique–it doesn’t require hospitalization or open surgery,” says physicist Gail ter Haar, head of therapeutic ultrasound at Royal Marsden Hospital in Sutton, England. Her group aims to make ultrasonic surgery a tool for treating cancer.
Most people encounter medical ultrasound with that first view of Junior in the womb. For years, doctors have also used ultrasound, as they do X-rays, to peer inside the body at bones, tumors, and other structures. They’ve even used it as a way to break up kidney stones and cataracts.
Now, technological innovations, including dramatic advances in computing power, have made more types of ultrasound treatment seem practical. Destroying malignant tumors and stopping the internal bleeding of trauma victims are among the first potential applications in the emerging field of ultrasonic surgery.
“A large number of folks who are subject to blunt trauma–they’re near an explosion, they’re in an auto accident, they’re Princess Diana–die of an internal hemorrhage . . . in 15 to 30 minutes,” says Kirk W. Beach, a surgeon at the University of Washington in Seattle. He and his colleagues at the university’s applied physics lab aim to preempt such tragedies by developing an ultrasound tool that could rapidly pinpoint and seal hemorrhages deep inside the body.
A silent punch
Ultrasound packs a silent punch. Pressure-waves at frequencies too high for people to hear emanate from a source, or transducer, much as ripples spread from the point where a rock drops into a pond. Ultrasonic transducers create these waves when a ceramic element vibrates, producing unseen ripples that are millimeters or fractions of a millimeter apart.
Ultrasonic transducers for medical imaging operate at very low intensity, applying a power of about one-tenth of a watt per square centimeter. That’s just 10 percent of the average power of sunshine at Earth’s surface. By contrast, the ultrasound used for new experimental procedures in bloodless surgery relies on a transducer that applies as many as 3,000 watts per square centimeter to a target area.
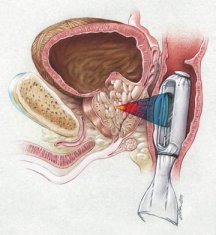
This tool, known as high-intensity focused ultrasound (HIFU), is analogous to a magnifying glass that concentrates enough sunlight to burn a hole in paper. A HIFU device quickly heats blood or tissue precisely where it’s focused. A dose lasting just a few seconds can cause targeted cells to die or can denature the proteins in blood so they congeal into a clot like the white of an egg does as it’s fried.
With the most sophisticated HIFU transducers, a researcher can focus the ultrasonic beam at a range of tissue depths and adjust its shape from circular to long and skinny. Even more-elaborate devices under development may eventually move the focus from side to side as well.
“HIFU was initially developed as a research tool to destroy regions of the cat brain for behavioral studies,” ter Haar says. Then in the 1950s, William J. Fry, a physicist at the University of Illinois at Urbana-Champaign, envisioned HIFU as a noninvasive surgical technique. With a grant from the Office of Naval Research, he, his brother Francis Fry, and Reginald C. Eggleton developed an experimental transducer system that successfully treated several people with brain disorders, particularly Parkinson’s disease. But with the development of the drug L-dopa for Parkinson’s, doctors turned away from surgical techniques.
In the 1960s, HIFU technology made further inroads as an experimental treatment when it was applied to glaucoma, an eye disease, but laser surgery superceded it. Now, after years of being overshadowed by other therapies, HIFU may finally become a treatment of choice.
Last year, the French company EDAP Technomed, based in Vaulx-en-Velin, sold to European hospitals the first commercial HIFU machines for noninvasive surgery. Seeking U.S. Food and Drug Administration approval for its device, designed to destroy prostate tumors, the company is conducting clinical trials on U.S. patients with prostate cancer. A second company, Focus Surgery in Indianapolis, will begin testing a similar device on cancer patients next month. Several other HIFU tools for scalpel-free surgery also are in the works, including ones central to the projects at the University of Washington and Royal Marsden Hospital.
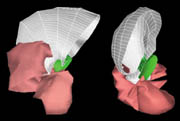
One of the biggest technical challenges of developing HIFU surgery, scientists say, has been finding a way to watch without a delay what the ultrasound tool is doing. Ideally, they want to create a tool that combines two ultrasonic devices: a HIFU transducer to carry out the medical process and an imaging transducer to watch the procedure as it unfolds. The intense HIFU waves overwhelm a standard ultrasound image and send streaks of light across the screen.
To overcome this problem, Beach’s colleagues at the University of Washington wired their HIFU and imaging transducers so the two fire in rapid alternation. During the period when their activity doesn’t overlap, the imaging device can show where the HIFU beam is having an effect. Although this solution sounds simple, it’s been “the most interesting and most challenging component of our work recently,” says Shahram Vaezy, a leader of the project.
Ter Haar says her group at Royal Marsden Hospital is also attempting to integrate HIFU with ultrasonic imaging to create a new device for treating liver cancer. The team should have a prototype by fall 2001, she says, but she declines to give details while the product is under development.
Her team has already tested HIFU on 50 patients with liver, prostate, and kidney tumors. In those cases, the researchers relied on magnetic resonance imaging, or MRI, to monitor the treatment before and after, but not during, the procedure, according to ter Haar. Because MRI machines are more costly, ultrasound imaging would be preferable, she says, especially “for small hospitals that don’t have MRI.”
Emergency situations
Emergency situations make the case for a combined HIFU-imaging device even more compelling. If an accident happens far from a hospital, for instance, the only hope of saving the victim’s life may be to stop internal bleeding in the field. With a portable HIFU-imaging tool, paramedics could detect and heal internal wounds. The University of Washington group is working toward a device that will help a paramedic do precisely this.
Given the sequence of decisions an emergency medical team might face, the scientists aimed for a quick way to diagnose internal bleeding, determine the rate of blood loss, and pinpoint the hemorrhage.
The technical challenges of detecting bleeding vary depending on the location of a hemorrhage, Beach says. His group is first tackling the problem of abdominal bleeding.
A pool of blood in the abdominal cavity looks like a dark crescent on an ultrasound screen. Beach’s recently graduated doctoral student Jong-Tae Yuk devised an ultrasound method to measure changes in blood volume in the abdomen once that ominous crescent appears. Existing ultrasound devices can’t make this measurement, according to Beach.
“Ultimately, [our machine] should give you a readout of the rate of blood loss,” he says, so even someone with little training in ultrasound can determine how quickly a victim needs treatment. “We want it to be as easy as using a cell phone,” he explains.
If an injured person were losing blood rapidly, a rescue crew would want to find the source of bleeding immediately and stop it with HIFU. In the abdomen, the source could be the liver, the spleen, or a blood vessel. The University of Washington group has developed an ultrasonic tool to pinpoint vascular bleeding and is working out techniques for spotting liver and spleen hemorrhages, as well.
“If there is bleeding from an artery into the [abdomen], there will be a vibration of the tissue where the bleeding occurs” due to eddies in the stream of blood spurting from the vessel, Beach explains. “We have modified conventional ultrasound imaging systems to look for vibration.”
The equipment is sensitive enough to detect movements of a mere 50 nanometers–less than a thousandth of a hairbreadth. A statistical program determines the probability that the given vibration does, in fact, indicate bleeding, and a color monitor displays its location.
Producing these pictures instantaneously demands a staggering amount of computer-processing power. “It takes a day for a desktop computer to make one image,” Beach says, and the team’s prototype will generate 30 images per second. A specialized computer chip would make this feasible, he says.
In laboratory experiments, the scientists have stopped bleeding in pigs’ livers with an early version of their HIFU-imaging device. Although the group focuses on abdominal bleeding now, it may eventually develop an ultrasound tool for spotting and sealing hemorrhages around the heart and brain, Beach says.
Cancer researchers also are eager to expand their applications of HIFU surgery to more organs. Oncologists may someday be able to routinely destroy tumors not only of the prostate, but also of the liver, kidney, breast, and brain.
Nearly a half-century after William Fry proposed HIFU surgery and more than 30 years after Star Trek depicted Dr. McCoy treating a patient wih it, ultrasonic surgery has a good shot at coming into its own. With over a million people in the United States developing cancer each year and thousands more dying annually of internal bleeding after a trauma, the advances of this technology could alter many fates.





