Cooking Up a Carcinogen
Should we worry about all that acrylamide in our diet?
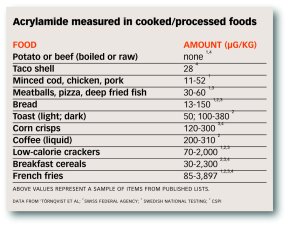
Turns out that hamburgers have it. So do french fries, crackers, breakfast cereals, pizza, fried fish, cauliflower au gratin, minced chicken, cooked beets, potato pancakes, powdered chocolate, and coffee. It’s acrylamide. Though best known as a carcinogenic and neurotoxic building block of many plastics, scientists are now discovering it in more and more foods. Oddly enough, it seems to be getting


there not as a chemical contaminant but as the product of common cooking practices. The toxic compound forms during chemical reactions between ingredients in a wide variety of foods as they fry, bake, or undergo other forms of heating.
Four months ago, scientists at the Swedish National Food Administration in Stockholm and Stockholm University announced the first detection of acrylamide in a host of fried and baked goods, especially potato chips and french fries (SN: 5/4/02,
p. 277: Available to subscribers at Cancer Link Cooks Up Doubt: Heating may form potential carcinogen in food). Since then, researchers elsewhere have launched a feverish campaign to identify what other foods carry the chemical.
Already, labs in several other countries have confirmed the Swedish findings. To date, of the 100 or so foods tested, several dozen appear to routinely cook up the carcinogen.
Scientists don’t know why acrylamide forms in food or how it’s escaped notice for so long. Nevertheless, as a moderately potent carcinogen in rodents, its apparent prevalence in the food supply suggests that it could pose a threat. Then again, junk-food addicts who down mountains of chips don’t appear to be succumbing in droves to cancer nor suffering tingling fingers and toes—the classic symptoms of acrylamide neuropathy.
This has led some toxicologists to question whether body processes disarm the poison. After all, Homo sapiens may have been eating acrylamide since people first tamed fire.
But, prompted in part by a June directive from the World Health Organization in Geneva, food scientists aren’t taking any chances. They’re developing an international research network to investigate the new concern and determine whether it’s much ado about nothing or a serious public health problem.
Crippled cows
Cooking’s creation of acrylamide might never have been revealed were it not for construction of the Hallandsås railway tunnel in southern Sweden. Blasted through a rocky ridge, the tunnel developed water leaks in 1997. Crews attempted to repair the fractures by pumping in an acrylamide-based liquid that hardens into a waterproof plastic.
Much of the liquid failed to harden, however, and instead seeped with the leaking water into an adjacent river. The acrylamide killed fish there and paralyzed cows drinking tainted water.
Concerned about the tunnel crewmembers who had been exposed to the flowing acrylamide for 2 months, Margareta Törnqvist and her colleagues at Stockholm University surveyed the workers’ blood for telltale markers of the poison. And they found them: fragments of acrylamide bound to hemoglobin in the workers’ red blood cells.
To the scientists’ amazement, they found the same acrylamide fragments on hemoglobin from people with no known exposure to the chemical. Though concentrations of altered hemoglobin in these volunteers were only a few percent of those in the workers, Törnqvist worried that they might reflect exposure in the general population.
Bound chemical fragments, so-called adducts, pose no apparent hazard on hemoglobin, Törnqvist emphasizes. But as markers of exposure to a chemical, they signal that a person’s DNA has probably acquired the same adducts. Unless repaired, DNA adducts can cause genetic damage and cancer. Indeed, by damaging DNA, acrylamide causes cancer in animals.
Acrylamide forms when tobacco burns, but that observation didn’t explain the acrylamide adducts in the nonsmoking volunteers. So, the Stockholm scientists decided to explore other high-temperature processes, such as cooking, as possible sources of the adducts.
Eden Tareke, for example, fed some rats fried food. She added water to dry, protein-rich rat chow that contained no acrylamide and shaped the doughy material into thin pancakes. She let some air dry at room temperature and fried the rest for 2 to 5 minutes. Tareke then provided each preparation to a group of rats for 2 months.
In June 2000, Tareke, Törnqvist, and their colleagues reported that the fried pancakes contained 100 to 200 micrograms of acrylamide per kilogram of chow and that the rats eating them developed acrylamide adducts on hemoglobin. Rodents eating unfried pancakes had only one-tenth that concentration of acrylamide adducts.
Test recipes
To investigate whether acrylamide taints people’s diets, the Stockholm group then looked at hamburger, which before cooking showed none of the chemical. “At that point,” Törnqvist explains, “we thought [acrylamide] formed from protein” when it’s cooked, as does another class of carcinogens, heterocyclic amines (SN: 4/24/99, p. 264: http://www.sciencenews.org/sn_arc99/4_24_99/bob1.htm). Sure enough, the researchers found that frying introduced 14 to 23 µg of acrylamide per kilogram of burger.
For a test of low-protein food, Tareke turned to potatoes. To her surprise, the undetectable amount of acrylamide in the raw vegetable rose to as much as 200 µg/kg in mashed potatoes, 660 µg/kg in french fries, 780 µg/kg in hash browns, and 3,800 µg/kg in chips.
Törnqvist’s group also tested fish, poultry, and pork. Again, all cooked up acrylamide—but typically only some 10 to 50 µg/kg. In April, the Swedish National Food Administration announced some of these results and its own data on potatoes, breads, cereals, and various fried foods.
The university scientists next tested other starchy foods and more cooking methods. They found that baking creates acrylamide in breads and crackers. Microwaving left potatoes with plenty of acrylamide, though boiling appeared to generate none. A full report of the provocative findings appears in the Aug. 14 Journal of Agricultural and Food Chemistry.
One of the biggest impacts of the April announcement was that it jump-started research by others, such as chemist Michael W. Pariza of the Food Research Institute at the University of Wisconsin–Madison.
For the past 4 months, he’s been looking for acrylamide in cooked foods, too. “And indeed, as have others, we found that it’s there,” he says. “What we now need to know is: Does it pose a risk, and is there anything we can do to limit it?”
Does it matter?
Robert G. Tardiff knows acrylamide well. Water-treatment plants add the chemical to drinking water because it precipitates out contaminants. Tardiff was the senior toxicologist in the Environmental Protection Agency’s Office of Drinking Water during the 1970s, when it established a safe concentration of the chemical, 1 part per trillion in water leaving plants.
Most of the toxicity data on which his group had based its regulation came from animals that had inhaled acrylamide, as a plastics worker might. However, those data probably produced a conservative limit. Tardiff explains that for most compounds, “you get more chemical per unit bodyweight into the bloodstream from inhalation than from ingestion or [skin] contact.”
Tardiff is now a consultant under contract to the Snack Foods Association of Alexandria, Va. Suggesting that acrylamide in food may be harmless, he says that other compounds in the diet might bind acrylamide, neutralize its toxicity, or reverse any adverse effects.
What’s more, he says, the hemoglobin adducts that researchers such as Törnqvist have found could be a good thing. Blood adducts “might well be a previously overlooked means by which the body keeps acrylamide away from sensitive tissues,” he says.
Hemoglobin circulates in the body for only 120 days before it—and any adducts on it—would disappear, Tardiff explains.
Overall, substances that cause cancer in rodent tests frequently haven’t been linked to cancer in people, notes Lois S. Gold, director of the Carcinogenic Potency Project at the University of California, Berkeley. In many cases, she suspects, rodent cancers may be triggered only by high experimental doses, which people never encounter.
Over the past 2 decades, she’s analyzed roughly 6,000 animal studies to compile a database on some 1,500 chemicals. More than half of the compounds caused cancer in rodents.
Gold says that against the enormous background of chemicals that people encounter, any single rodent carcinogen is not likely to be an important source of human cancer. Why? Perhaps typical doses of chemicals that people take in are too small to cause cancer or the mechanisms that trigger cancer in rodents don’t operate in people. Nevertheless, she maintains, data on the potency of rodent carcinogens could set priorities for which compounds most warrant investigation into their mechanisms of action.
Toward that end, her group has developed a carcinogen potency index, the HERP index, which represents human exposure divided by rodent potency.
Prompted by the new Swedish estimate that people consume perhaps 40 µg of acrylamide per day, Gold has calculated a HERP score indicating that such a human intake is 0.015 percent of the body-weight-adjusted dose that gave cancer to 50 percent of exposed rats. She notes comparable HERP scores for average U.S. intake of several known rodent carcinogens: catechol and furfural from coffee, hydrazine from mushrooms, and caffeic acid from lettuce. Those figures are well below the HERP score for exposures to formaldehyde and ethanol, Gold emphasizes.
That’s not to say that people should dismiss acrylamide, Gold says. The chemical’s HERP score, in fact, is somewhat higher than average for all the rodent carcinogens analyzed so far. So, she too welcomes more research into acrylamide’s biological actions.
What’s a diner to do?
Until acrylamide’s human risks are sorted out, Food and Drug Administration Deputy Commissioner Lester M. Crawford says there’s no reason people should change their diets, as long as they’re following FDA’s current recommendation to “eat a healthy, balanced diet consisting of a wide variety of foods from a variety of sources.”
Indeed, the Washington, D.C.–based American Institute for Cancer Research notes that the foods with the highest acrylamide concentrations—potato chips and fries—”are high-fat, high-calorie, nutritionally poor options” that “have been convincingly linked to greater cancer risk.”
Michael F. Jacobson, executive director of the Center for Science in the Public Interest, a public-advocacy group also based in Washington, D.C., agrees that acrylamide gives people yet another reason for “cutting back on their french fry consumption.” When his center sent samples of fast foods and grocery items to Swedish researchers, the tests confirmed that acrylamide is present in U.S. foods.
Rather than attempt to cull from the diet every food linked to acrylamide, “we should aim to eat less of the most contaminated, least nutritious foods,” says Jacobson. “For instance, I wouldn’t give up eating something like Cheerios, even though our data show there’s a little [acrylamide] in there,” he says. “It’s still basically a healthful, whole-grain, low-sugar food.”
Jacobson’s assessment echoes many others: Acrylamide in food “isn’t something that people should panic about.”







