Daily drug shown effective in preventing HIV infections
Additional preexposure prophylaxis options now under development
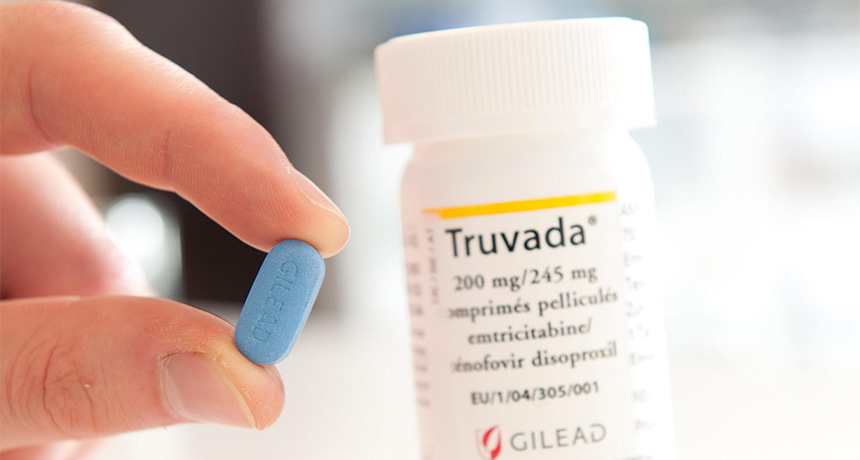
AT RISK Recent studies have found that Truvada, a drug to prevent HIV infection in people at risk of exposure to the virus, is effective when taken daily.
© EPA European Pressphoto Agency B.V./Alamy Stock Photo