Richard Kriwacki refused to give up on his protein. He had tried again and again to determine its three-dimensional shape, but in every experiment, the protein looked no more structured than a piece of cooked spaghetti.
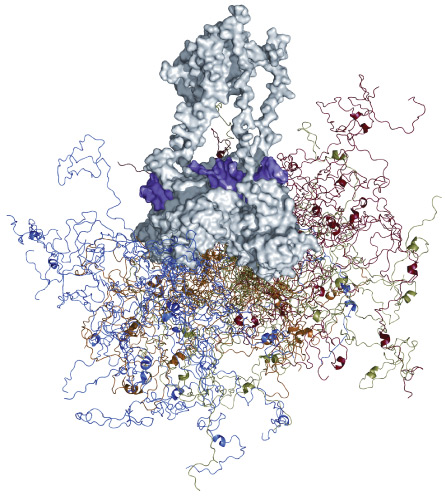
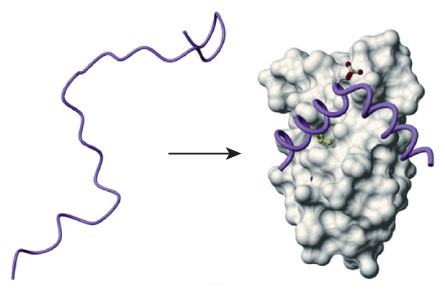
Normally, this lack of form would be a sign that the protein had been destroyed, yet Kriwacki knew for a fact it could still do its job in controlling cell division. While discussing the conundrum with his adviser in the atrium of their La Jolla, Calif., lab, insight dawned: Maybe the floppy protein didn’t take shape until it attached to another protein. Kriwacki raced off to do yet another experiment, this time combining his protein, p21, with a partner. Sure enough, Kriwacki got what he was looking for. Once joined, a seemingly ruined mess gave way to a neatly folded structure. The finding defied a foundational dogma of biology, that structure determines function.
Nearly everything the human body does, from shuttling oxygen through the bloodstream to digesting a meal, relies on proteins. These biological workhorses are composed of chains of molecules called amino acids. Whenever a chain is made, conventional scientific wisdom says, electrical forces cause it to immediately bend into helical ribbons and tight zigzags, which twist further into even more defined three-dimensional forms. The resulting shape determines what other molecular players the protein can bind to and thus what it can accomplish in a cell. Unfolded proteins were thought to result only from intolerable conditions that render a protein useless, such as extreme heat or acidity.
But since around the time of Kriwacki’s discovery more than 15 years ago, disorder has surfaced as a key player in the protein world. “Intrinsically disordered proteins,” or IDPs, turn out to play vital parts in controlling cellular processes. More than one-third of all human proteins, in fact, may be partially or completely disordered in structure, floating around like strands of wet noodles. “The roles that disordered regions can play are quite diverse,” says Kriwacki, now at St. Jude Children’s Research Hospital in Memphis, Tenn.
To better understand how something so flexible can be functional, researchers are now taking a closer look at how the disordered proteins interact with other proteins. The disordered dissidents can behave as switches, quickly turning cellular processes on or off in response to changing conditions, or as shape-shifting ensembles that integrate multiple signals before telling a cell to get a job done. Studying the interactions of intrinsically disordered proteins may even yield insight into certain diseases and lead to new treatments.
Floppiness exposed
Disordered proteins flew under the radar for so long because the standard technique for determining a protein’s structure, known as X-ray crystallography, requires that the protein retain a set shape long enough to be crystallized. Scientists had found a few examples of proteins that couldn’t be crystallized, but these were thought to be anomalies.
When Kriwacki encountered the troublesome p21 protein, he was working with molecular biologists Jane Dyson and Peter Wright at the Scripps Research Institute. Dyson and Wright were using a technique called nuclear magnetic resonance, or NMR, spectroscopy, which reveals a molecule’s form based on the magnetic properties of its atoms’ nuclei as opposed to its crystal structure. “Peter and Jane’s lab at the time was the world-leading protein NMR lab,” says Kriwacki. Advances in NMR were what allowed him to finally figure out what his protein looked like.
After p21, examples of these proteins just kept turning up. You don’t need to go looking for them, Dyson says, “they’ll come looking for you, believe me.” In 1999, Dyson and Wright published a landmark review paper in the Journal of Molecular Biology that set the stage for a new protein paradigm. There were too many examples to be mere outliers; it was clear that something bigger was going on. “We were finding that these proteins were not only unstructured, but had to be,” Dyson says.
Meanwhile, other scientists were independently building a strong case for the existence of intrinsically disordered proteins. Keith Dunker, a bioinformatician at Indiana University School of Medicine in Indianapolis, and Peter Tompa, a protein chemist at the Free University of Brussels, were both leading efforts to predict disorder mathematically. “The main thing we did was to determine that unstructured proteins have a fundamentally different amino acid composition compared to structured proteins,” Dunker says.
A protein’s mix of amino acids can create regions that are either hydrophilic (“water-loving”) or hydrophobic (“water-hating”). Structured proteins that exist in solution typically fold into spherical shapes with a hydrophilic shell enclosing a hydrophobic core. But disordered proteins contain few, if any, hydrophobic regions, so they don’t fold up. They also tend to have more electrically charged portions. “If you look at these differences, you can anticipate that they’re not going to fold into a 3-D structure,” Dunker says. To help study the differences, he and his colleagues developed “DisProt,” a database of proteins that experiments have shown to be disordered.
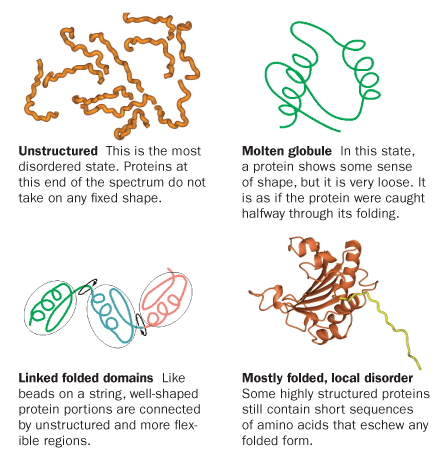
Though scientists often speak of “structured proteins” and “intrinsically disordered proteins” as if they are distinct classes, along the way it has become clear that any particular protein’s degree of disorder falls on a spectrum, from precise rigidity to complete disarray. Proteins can also migrate along that spectrum from one moment to another, shifting into different versions of themselves. Many disordered proteins, including p21, eschew their wiggly nature when binding to a partner protein — like a string puppet snapping to attention. Others fold to a more limited extent upon binding, and some never shape up at all.
Fold for a cause
How a protein’s degree of disorder enables its function is now a hot topic of research. Ongoing efforts suggest some disordered proteins act like switches, triggering or stopping an action in response to a signal. This makes them well-suited for controlling activities such as the production of other proteins, cell growth or division, and the sending of cellular signals.
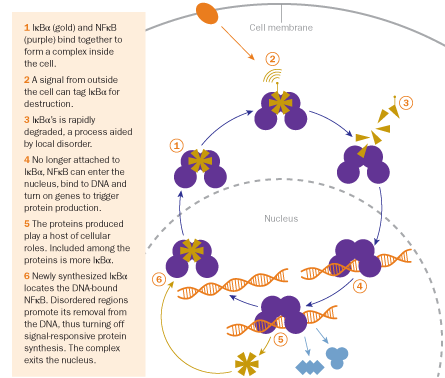
Lately, Dyson has been working with Elizabeth Komives of the University of California, San Diego to study a duo of proteins, NF-kappaB and I-kappa-B-alpha. Together, the proteins control a host of vital phenomena in cells, from growth and development to immunity and stress response. The proteins, which both contain disordered regions, normally exist bound together as a complex within a cell. When the cell receives a signal, such as a hormone molecule binding to its surface, IκB± is tagged for destruction and degraded. NFκB is released and sent to the nucleus. There, NFκB binds to the DNA, turning on genes that hold the instructions for making specific proteins.
One of the proteins produced is more IκB±, which allows the response to be switched off again when it is no longer needed. IκB± binds to NFκB and strips it from the DNA, Dyson and Komives reported last year in IUBMB Life. It’s not yet clear how the stripping process works, but the disordered regions of IκB± appear to cast around like a fishing line to find NFκB and peel it off the DNA. The new complex of NFκB and IκB± leaves the nucleus and returns to its resting state within the cell. Thanks in part to the disordered regions, the cell can respond flexibly and rapidly to external stimuli.
While NFκB and IκB± become mostly structured upon binding, other IDPs remain highly dynamic. One example is Sic1, a disordered protein found in yeast that prevents DNA replication and thus keeps cells from dividing. A 2008 study led by Julie Forman-Kay of the University of Toronto and then-colleague Tanja Mittag revealed how proteins such as Sic1 function as “dynamic ensembles” of disordered states. Sic1 contains six short disordered regions that take turns binding in a “pocket” of a partner protein. At any given moment, only one of Sic1’s six regions sits inside the pocket, while the other regions remain disordered. Each of these six regions is susceptible to modifications that can deactivate it in a way that prevents it from binding. All six of them must be ready to bind for Sic1 to hold onto its protein partner and stave off cell division. Each segment’s activation is like a weight added to one side of a balance — only with enough weights does the scale tip.
While some disordered regions play active roles in sensitive responses, others serve only to hold more structured areas together like beads on a string. The disordered protein complex CBP/p300 has several structured regions connected by long, floppy “flexible linkers.” The linkers form a malleable scaffold for bringing together the structured parts of the protein complex, controlling how and when these other players interact.
In sickness and health
Historically, before IDPs took off, anything other than a properly structured protein was considered a disease risk. This was a reasonable conclusion, given that diseases often result when proteins take forms they aren’t meant to, a process called misfolding. Today, though, scientists know that a disordered protein is not the same as a misfolded one.
Still, IDPs, like any proteins, can misfold. And misfolded proteins known to play roles in some high-profile diseases have recently turned out to be disordered. The tau protein, for example, forms the characteristic protein tangles seen in Alzheimer’s disease. Same, it seems, for alpha-synuclein and Parkinson’s disease. Some scientists think disordered proteins may be more prone to misfolding than other types, but the relationship is not yet clear. By understanding the full range of protein folding behavior, scientists hope to gain insight into the causes of such diseases.
Homing in on interactions involving disordered proteins could also lead to new approaches to treatment. Drug developers have traditionally focused on creating molecules that bind to highly structured proteins that carry out reactions in a cell. That means binding to what’s called an “active site.” But the new understanding of IDPs opens possibilities for designing drugs that instead interfere with protein-protein interactions, by binding to intrinsically disordered proteins or binding to a site on a partner protein where the IDP attaches.
“The idea of targeting disordered proteins themselves remains very challenging,” Kriwacki says. “Much more feasible is to target binding sites on folded [partner] proteins.” If a short sequence of a disordered protein is known to bind to another protein that triggers a disease state, a small molecule could mimic that sequence, binding to the partner protein and deactivating it. An anticancer drug developed by the pharmaceutical company Roche is made from Nutlin-3a, a chemical that works in just this way. Nutlin-3a prevents an IDP commonly associated with cancer, p53, from interacting with its partner protein.
Of course, scientists’ current understanding of disordered proteins assumes that the versions studied in lab dishes are in fact disordered in cells, a notion some researchers challenge.
Disorder doubters
As with any paradigm shift, the idea that proteins can be disordered but still functional has its skeptics. “I think the majority of people accept disorder,” says Wright, “but there are still a few critics.” Most studies of IDPs are conducted in lab dishes rather than in living cells, because today’s techniques, for the most part, aren’t sensitive enough to allow the study of proteins at the low amounts present in actual cells. Scientists commonly use bacterial cells to create many copies of a protein. The protein is then isolated and studied under artificial conditions. This has led some researchers to question whether the apparently disordered state of IDPs is merely an artifact of the lab environment. In the true setting of a cell, which is much more crowded with other molecules, the proteins might be folded, the critics argue.
Neuroscientist Dennis Selkoe of Harvard Medical School and colleagues published a controversial paper in 2011 suggesting that alpha-synuclein protein, widely believed to be disordered in its healthy form, actually exists in a structured state inside cells. Selkoe’s team studied alpha-synuclein obtained from human brain cells grown in a lab dish, reporting that the protein appears to occur naturally as a helix-shaped “tetramer” of four proteins as opposed to a single, unstructured protein.
But the findings are highly contested, and others have failed to replicate them. Philipp Selenko, a biochemist at the Leibniz Institute of Molecular Pharmacology in Berlin, used NMR to show that alpha-synuclein was unstructured inside intact E. coli bacteria. Biologist Guy Lippens of Lille University of Science and Technology in France and colleagues have shown that tau protein, too, appears unstructured in immature frog egg cells. Still, says Lippens, the question of whether all lab-studied IDPs are truly disordered in cells remains open.
Assuming the proteins are unstructured, another mystery is how they evade degradation. Cells have machinery that recognizes proteins that haven’t folded properly and digests them. One theory posits that since IDPs lack regions of the type the degradation system recognizes, the disordered proteins appear to the cell as folded proteins. Another theory holds that “chaperone” proteins bind to IDPs to stabilize them so they don’t get eaten up by the cell. A third theory suggests that IDPs are tightly regulated and kept at low levels in the cell, broken down when they are not needed. Studying proteins under natural conditions — in cells — will help provide the answer.
Despite a growing awareness of disorder in proteins, much more research remains to be done. In this chaotic new view of the protein world, scientists must reexamine everything they have assumed about structure and function. “Just like in physics,” Tompa says, “the protein universe seems to have this dark matter we have neglected, which now turns out to be important in cells.”
Loose jobs
Intrinsically disordered proteins, or IDPs, have important regulatory and signaling jobs in cells (some outlined below). Their disorder is thought to make them better at these tasks, by enabling quick and flexible responses to the changing conditions that cells face.
- Cell cycle activities Disordered proteins help control when and how a cell grows and divides.
- Transcription These proteins turn on and off the copying of genes (DNA) into protein-making instructions (RNA).
- Translation IDPs are involved in the reading of RNA to make proteins.
- Signal transduction The flexibility of intrinsically disordered proteins allows them to convert a signal coming from outside the cell into a response that shows up within the cell.
- Self-assembly of multiprotein complexes IDPs help bring together different proteins to form larger structures — such as the ribosome, a molecular machine that serves as the site of protein synthesis.
- Cargo transport These proteins play a role in moving large molecules around a cell along the fibers making up the cell’s skeleton.
- Apoptosis Disordered proteins can mediate a cell suicide pathway.






