Doctors enlisted to turn the tide on antibiotic resistance
Behavior change — among prescribers and patients — can combat resistant microbes
LOW-TECH BACTERIA BATTLE Clostridium difficile (shown) is common in hospitals and is hard to treat.
CDC
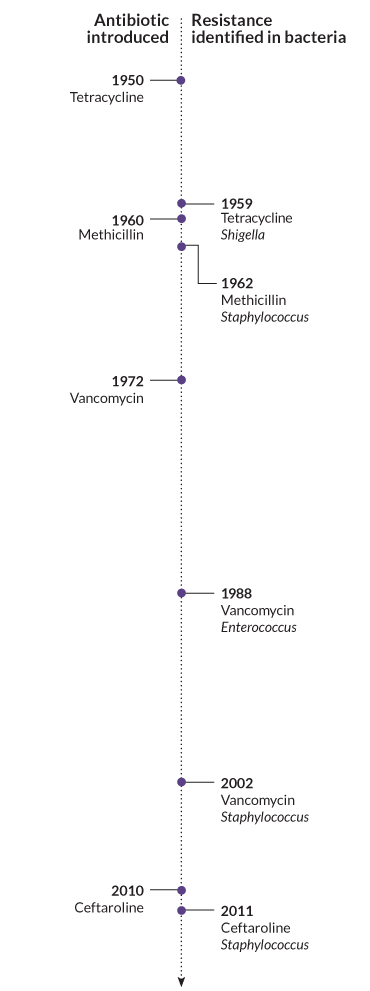
It is not difficult to make microbes resistant to penicillin in the laboratory by exposing them to concentrations not sufficient to kill them….
—Alexander Fleming, discoverer of penicillin, in his 1945 Nobel Prize lecture
Fleming’s remarks were spot-on. Since the heady days of penicillin’s discovery, an overuse of antibiotics has spawned bacterial resistance to the drugs and threatened to erase decades of success. Every prescription that misses the mark or throws excess drugs at a bacterium gives bystander bacteria a good look at those antibiotics and a head start in resisting their effects, as Fleming noted.
Some microbes are changing faster than antimicrobials can kill them. As a result, it’s once again possible to get a bacterial or fungal infection for which there is no sure cure. That’s how roughly 23,000 people die in the United States each year.
As hopes fade that new antibiotics will save the day, many treatment centers are taking a surprisingly low-tech approach: They are changing the behavior of prescribing physicians. The goal of this tactic, called antimicrobial stewardship, is to curb antibiotic resistance by speeding diagnoses, getting the most appropriate drug to each patient, limiting intravenous drug delivery and avoiding use of broad-spectrum antibiotics — scattershot drugs that hit more than one kind of microbe. Evidence shows that antimicrobial stewardship can shorten a patient’s hospital stay and lower drug expenses. Fighting resistance may not be futile.
Antimicrobial stewardship requires an agile response from institutions that are not known for changing quickly. But it mainly demands human investment: training, diligence and doctors’ agreement to abide by guidelines and relinquish some control over prescribing. Consumers also have to do their part. The days of demanding antibiotics at the doctor’s office may soon be history.
It won’t be easy. Ending antibiotic resistance, or even getting the upper hand, will take more than antimicrobial stewardship: It will demand the relentless use of infection-control measures such as hand-washing and “gowning and gloving” in hospitals and clinics. Pharmaceutical companies will need to develop new antibiotics. Plus, doctors will need prompt access to top-line diagnostics to identify ever-changing microbes (see sidebar), the high-tech piece of the puzzle.
In the early antibiotic era following World War II, a stream of drugs from Big Pharma kept bacteria at bay. But the pipeline is running dry. “Pharma is not producing antibiotics at a rate that allows us to stay ahead of the problem,” says physician Scott Flanders of the University of Michigan in Ann Arbor. There’s not much profit in creating drugs if bacteria are just going to outsmart them. The deck seems stacked against a supply-side solution.
Some new drugs are likely to get approved in coming years, but prescribing behavior will have to change to take advantage of them, says physician Dennis Maki of the University of Wisconsin–Madison. “The development of new antibiotics without having mechanisms to ensure their appropriate use is much like supplying your alcoholic patients with a finer brandy.”
Antibiotic overload
Well-known bacteria such as Staphylococcus, Streptococcus, Escherichia coli, Salmonella and Neisseria gonorrhoeae are increasingly dodging the effects of drugs as are other less-known but dangerous microbes, the World Health Organization reported in April. Antibiotic resistance frequently shows up in patients with tuberculosis, pneumonia, wounds and urinary tract infections.
Resistant infections send doctors searching for another pill that will work. More than half of patients discharged from U.S. hospitals received antibiotics during their stay in 2010, the U.S. Centers for Disease Control and Prevention estimated in a report released in 2014. The report suggests that antibiotic prescriptions for some ailments could be cut by more than one-third with key interventions.
“Our goal is not in any way to limit necessary antibiotic use,” says study coauthor Arjun Srinivasan, a CDC physician. “We are seeking to improve the use of antibiotics so they remain effective for patients who need them down the road.”
The kind of controlled use CDC is aiming for would logically start in hospitals, especially in the intensive care unit — resistance central. Patients who land in the ICU are already in trouble, and doctors tend to medicate them thoroughly. ICU patients have the highest antibiotic use in hospitals, says Nick Daneman, an infectious disease physician at the University of Toronto.
Those patients also have the highest resistance rates, he says. For example, bloodstream infections in patients, which can trigger a lethal condition called sepsis, were traceable to multidrug-resistant pathogens in 23 percent of patients analyzed at nine hospitals in the southeastern United States, according to a report in the March 18 PLOS ONE.
Antibiotics taken unnecessarily are not harmless, says physician Jeffrey Linder of Harvard Medical School. He points to Clostridium difficile as an example of what can go wrong. C. diff, for short, is a bacterium that can establish itself in the gut and cause debilitating diarrhea when it overgrows. Good microbes that dwell in the intestines normally suppress levels of C. diff, but the microbe circulates in hospitals and strikes patients who are on antibiotics that have wiped out some of those protective gut bacteria. When C. diff takes hold, it’s hard to treat (SN Online: 10/4/13).
To illustrate, physician Timothy Sullivan of Mount Sinai Hospital in New York City describes a recent case of C. diff in August’s JAMA Internal Medicine: A woman in her 80s with diabetes arrived at a hospital emergency room with an infected cut on her arm. She got three antibiotics and treatment for the wound. A soft tissue infection required surgery to remove dead tissue. She stayed in the hospital and received broad-spectrum antibiotics for three weeks as the surgical site healed. The day after her antibiotics were stopped, she developed a C. diff infection, with diarrhea, fever, dropping blood pressure and signs of kidney failure. Despite best efforts, she died.
People entering a hospital with an indeterminate-but-serious infection often get antibiotics immediately because, if the infection really is bacterial, the patient might die without the drugs, Flanders says.
CDC recommends a stewardship review of prescriptions within 48 hours after the first dose by an impartial source, such as an infectious disease doctor or pharmacist. More than second-guessing the prescribing doctors, these audits offer a chance to fine-tune the treatment if the diagnosis has changed, says Daneman.
He and his colleagues conducted a study of critical care patients in which they intervened as necessary three days after the first dose of antibiotics and again at day 10. The diagnosis for many patients had changed. A stewardship pharmacist spoke with the doctors attending the patient regarding antibiotic use. Doctors accepted 82 percent of those recommendations. As a result, broad-spectrum antibiotics use at the large hospital was reduced from earlier rates and, most importantly, ICU cases of C. diff originating in the hospital decreased. The study appeared in Infection Control and Hospital Epidemiology in 2012.
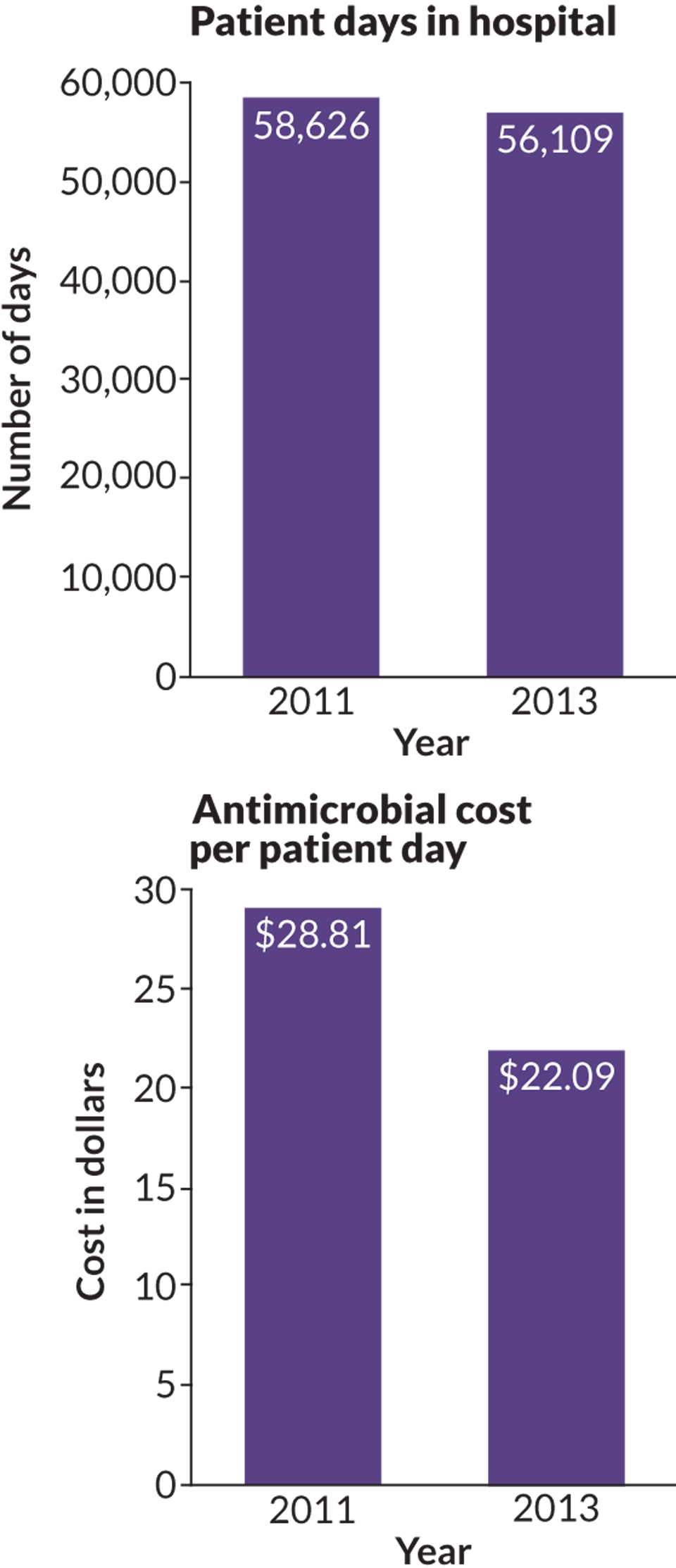
At Blount Memorial Hospital in Maryville, Tenn., pharmacist Brad Crane evaluates prescriptions and recommends changes. Prescribers accepted more than 90 percent of 977 recommendations made by Crane and colleagues in the first two years of the hospital’s stewardship program. Antimicrobial costs per patient have dropped from an average of about $29 to $22 per day, a sign of more efficient use of the drugs. Crane says the results show that stewardship can work at community hospitals, not merely at large facilities affiliated with universities. He reported early findings at a medical meeting in 2013.
Blount Memorial Hospital uses a strategy typical of stewardship programs. The aim is to steer doctors away from intravenous drugs, since IV lines can get infected, and discourage broad-spectrum antibiotics in favor of narrowly focused drugs.
“We tend to use antibiotics too long and at too-high doses,” says Neil Fishman, an infectious disease doctor at the University of Pennsylvania. Antibiotic stewardship can improve quality of care and cut costs, he says, so it’s not difficult to convince hospital administrators of the programs’ value. Flanders notes that hospitals are increasingly judged on how many hospital-origin infections they have. Any program that lowers that number is welcome, he says.
Outpatient clinics
Hospitalized patients are typically quite sick and many have bacterial infections. In those cases, the stewardship challenge is to determine the correct antibiotic. The question facing doctors in out-patient clinics is often different: “Does this patient need any antibiotic at all?” says Ralph Gonzales, a physician at the University of California, San Francisco.
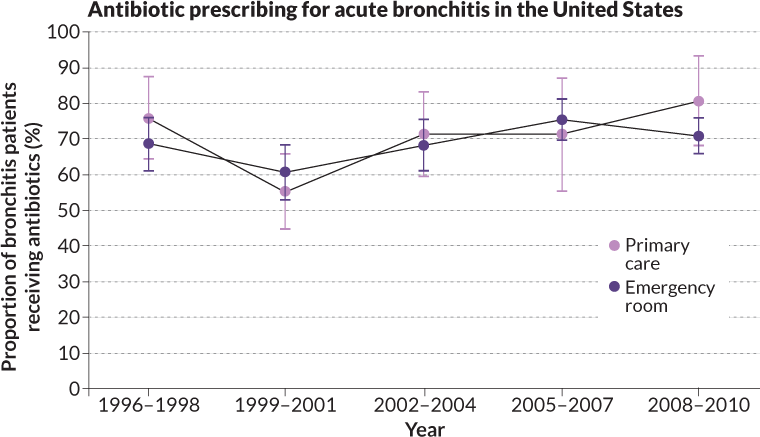
Outpatients walk in under their own power, often with nondescript symptoms they have researched on the Internet. Some ask for drugs up front. Bronchitis is a common complaint, and antibiotics typically don’t help because more than 90 percent of the time the illness is caused by a virus, says Linder, the Harvard physician. With or without drugs, he says, “it’s gone by day 20.” Even so, more than half of U.S. patients with bronchitis are prescribed antibiotics. “There’s this magical thinking that an antibiotic prescription will treat a viral infection.”
To test whether stewardship interventions can clean up prescribing in clinics, Gonzales and his colleagues enlisted 33 clinics in central Pennsylvania for a study. Eleven received a large poster for the examination room showing low-to-high likelihood of a patient having bacterial pneumonia — which would merit antibiotics — or a viral ailment. Eleven other clinics received decision-making guidelines electronically that doctors and nurses could consult when diagnosing respiratory infections. The last 11 clinics got neither.
Over six months, rates of antibiotic prescribing dropped from 80 percent to 68 percent in clinics showing the posters and from 74 percent to 61 percent in those getting computerized guidance. Prescriptions were largely unchanged in the control group, where about three-fourths of patients got antibiotics for respiratory infections, the researchers reported in 2013 in JAMA Internal Medicine. While that reduction might seem modest, Gonzales says, “across the population that accounts for a lot.”
Doctors’ chief concern when testing a patient with a respiratory ailment is pneumonia, he says. The electronic guidance helps them sort that out, he says, providing details on symptoms that point to pneumonia and away from myriad other respiratory ailments.
The posters help them counsel patients. Adults coming in with a cough-related illness often are in some kind of denial, Gonzales says. “I tell them this drug could do more harm than good, and they say, ‘That’s OK, I’ll take my chances.’ ” He adds, “[The poster] helps to provide some collective norms about proper antibiotic use.”
Going a step further, some researchers have asked prescribers to sign and post a commitment letter about antibiotic use in the exam room. Jason Doctor, a psychologist at the University of Southern California in Los Angeles, tested the effect of a letter stating that antibiotics can make bacteria more resistant and that getting them needlessly can cause problems.
“This was a powerful motivator,” he says. Having the letter visible in 14 exam rooms coincided with an average drop in inappropriate prescribing from 44 percent to 34 percent. The key was getting the doctors to sign and post the letter. It made them more accountable, Doctor says. “Precommitment ties you to the mast.” The report appeared in JAMA Internal Medicine in March.
Prescriptions in doctors’ offices are harder to track than in hospitals. But prescribers doled out antibiotics for sore throats in about 60 percent of adult cases between 1997 and 2010 in the United States, even though only about 10 percent of sore throats in adults are due to strep bacteria and require the drugs. In clinics, doctors face pressure “to appease the patient” even when an antibiotic isn’t needed, says Ohio State University pharmacist Debra Goff.
There are other pressures, too. “It takes a minute to write a script,” Fishman says. “It takes 15 minutes to not write one, to provide education.”
And even when strep throat is correctly diagnosed, Linder says, some doctors treat it with the broad-spectrum antibiotic azithromycin, sold as Z-Pak, to which Streptococcus pyogenes has shown resistance. Use of penicillin, to whichS. pyogenes has never been resistant, “has dwindled off,” he says. Z-Pak requires a shorter course than penicillin. People and doctors have this perception that newer is better, Linder says. “Z-Pak sounds cool.”
Turning the Titanic
Antibiotics deserve a big share of the credit for improved health care in the 20th century, but now their sporadic failure risks sabotaging those gains. Hospitals have changed from healing centers to risky stopovers where one is advised to get in and get out before catching a superbug. Not even Alexander Fleming could have predicted that.
The medical community was caught off guard. “We used to consider antibiotics as all value-added, not harmful at all,” says Sara Cosgrove, an infectious disease physician at Johns Hopkins Hospital in Baltimore. “Nobody really believed it would get to this point.”
It will take more than antimicrobial stewardship programs to reverse the trend. Resistance is a global phenomenon with antibiotics sold over the counter in many countries. In Mexico, Gonzales and his colleagues found that about half of 101 insured individuals who visited a family medicine clinic with a respiratory ailment were already on antibiotics, 20 of whom were self-treating. When interviewed, many mistakenly thought common cold remedies were antibiotics.
Ultimately, patients need to know better, Linder says, but he can sympathize with them. People often take a half-day off work to go to a clinic with a respiratory infection. “I spend 10 minutes with them, tell them there are no drugs and that they’ll be sick for two weeks. It’s all very unsatisfying,” he says. “No wonder the prescriptions get written a lot.”
Creative approaches might work. Gonzales and his colleagues used computer screens at kiosks in hospital emergency rooms to quiz people, in English or Spanish, on what they know about antibiotics. Of 686 people coming in with respiratory infections, only 22 percent initially doubted they needed antibiotics. That fraction rose to 49 percent after using the kiosk, the scientists reported in Patient Education and Counseling in 2011.
The key to establishing the value of such antimicrobial stewardship programs, whether aimed at doctors or patients, will be to show they actually slow or stop resistance. These data are just starting to trickle in. Daneman and his colleagues pored over 24 studies of stewardship programs and found that after being in place for more than six months, many showed a reduction in resistance, particularly in ICUs. The rate of decrease varied among microbes. And last month, at the Interscience Conference on Antimicrobial Agents and Chemotherapy in Washington, D.C., researchers at New York Hospital Queens in Flushing, N.Y., reported reduced rates of multi-drug-resistant Klebsiella pneumoniae, Acinetobacter baumannii and Staphylococcus aureus after two years of a stewardship program. The hospital is also seeing fewer readmissions within 30 days for infections, physician Nishant Prasad said.
Antimicrobial stewardship, widely applied, could curb resistance at the source, Fishman says. But wide-scale adoption will take time.
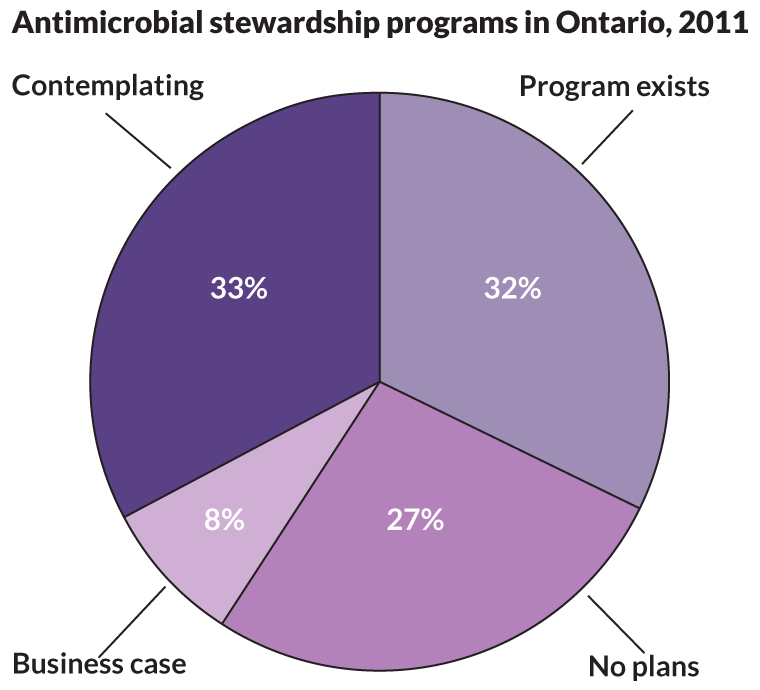
“We’re turning the Titanic,” Linder says. Even so, a 2012 survey by the Children’s Hospital Association found that 31 of 43 hospitals surveyed either had a stewardship program in place or were planning one. A nationwide survey of doctors found that about half worked in facilities with formal programs in place, and more than 90 percent were using some form of stewardship techniques. And a California survey revealed that half of hospitals there had a stewardship program and another 30 percent planned one.
The Obama administration appears ready to push the issue. At a July meeting of the President’s Council of Advisors on Science and Technology, panel cochair Eric Lander raised the possibility of requiring hospitals to have a stewardship program to receive Medicaid and Medicare
reimbursement. “We believe that having an antibiotic stewardship program is a reasonable expectation,” he said.
This kind of hammer is necessary, says Fishman, otherwise some doctors could continue to dish out drugs without regard for their macro effect on the microbe population.
“This really is a ‘tragedy of the commons,’ ” says Jason Doctor, the psychologist, referring to the classic tale of farmers overgrazing their animals on community land until it was lost to use. “Clinicians are using the public ‘space’ to prescribe big antibiotics. The potential harm to the greater good is devastating. We’re going to really be in trouble if we don’t manage this issue.”
Delays matter
A fast, accurate diagnosis can aid antimicrobial stewardship by curbing the drug-bug mismatches that add to resistance:
Polymerase chain reaction (PCR) Staphylococcus aureus hangs out innocently in most people, but it can also cause infection and gain resistance to the antibiotic methicillin. Ohio State University pharmacist Debra Goff and her colleagues tested 74 people at OSU Medical Center in 2008 using standard S. aureus diagnostics, which took about three days. In 2009, the researchers repeated the test on 82 patients, using PCR after the initial blood culture, to amplify the bacteria’s DNA and discern within an hour if a patient had methicillin-resistant S. aureus. Those patients got the right meds 1.7 days sooner on average than the 2008 group, the scientists reported in Clinical Infectious Diseases in 2010. “We saved $21,000 per episode of staph,” Goff says.
Mass spectrometry Researchers at Houston Methodist Hospital analyzed 265 patients with gram-negative bacteria infections. Patients treated before the researchers had mass spec, a rapid chemical analysis, waited more than 80 hours to get optimal antibiotic therapy. When mass spec was available, patients received the correct drug within 30 hours, as reported in the Journal of Infection in September. Only 9 percent of patients aided by mass spec died compared with 21 percent who got the slower diagnosis.
Procalcitonin test This peptide appears in the blood during bacterial infections, which can lead to sepsis, a lethal condition. At Mercy Medical Center in Des Moines, Iowa, researchers checked procalcitonin levels in 35 patients with suspected sepsis or pneumonia. Those patients, on average, needed only 10 days of treatment versus 14 days in a similar group that didn’t get the test, according to a 2013 report.
Rapid antigen detection test A quick test to distinguish strep throat from a viral sore throat is available in many clinics. It can rule out strep — and the need for antibiotics — in about 10 minutes. The test has the most utility in children, who are more prone to strep than adults. — Nathan Seppa
Editor’s note: After this story went to press, the Obama Administration on September 18 announced the launch of a five-year plan to fight antibiotic resistance. The effort will include expansion of antimicrobial stewardship programs in hospitals, clinics and long-term care facilities, public and private.