Medicated eye drops may delay nearsightedness in children
The earlier myopia starts, the worse eye health can become later in life
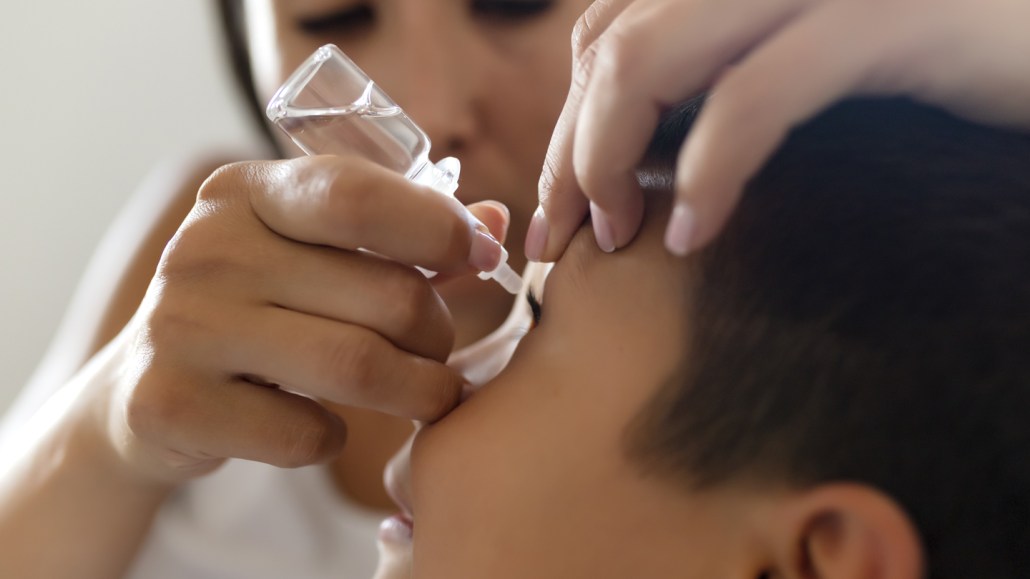
Medicated eye drops taken nightly for two years delays nearsightedness in children, a new study suggests.
PixelsEffect/iStock/Getty Images Plus
An eye drop a day could keep myopia at bay — at least temporarily.
Using nightly eye drops with 0.05 percent atropine, a medication that relaxes the eye muscle responsible for focusing vision, may delay myopia onset in children, researchers report February 14 in JAMA.
Myopia, also called nearsightedness, is an irreversible condition in which the eyeball grows too long front to back, causing blurred distant vision. It typically begins in childhood, and the earlier it starts, the worse eye health can become later in life. Elongated eyes increase the risk for ocular complications including cataracts, glaucoma and macular degeneration.
The prevalence of myopia has risen rapidly over the last few decades. About one-fourth of the global population currently has the condition. It is expected to affect half of people worldwide by 2050.
Genetics plays a large role in the condition. A 2020 study found that myopia risk is more than 10 times as high in children of two highly myopic parents as in children of nonmyopic parents, says ophthalmologist Jason Yam of the Chinese University of Hong Kong.
But he and other scientists speculate that environmental factors such as less time outdoors and more intensive education are causing the recent boom (SN: 1/24/13). “It’s happening too quickly to be a purely genetic or inherited issue,” says optometrist Kathryn Saunders of Ulster University in Coleraine, Northern Ireland, who was not involved in the new study.
Low-dose atropine eye drops are already used to slow myopia progression in several countries in Asia. Yam and colleagues wanted to see if the medication could also delay myopia onset.
The team recruited nonmyopic children ages 4 through 9 who lived in Hong Kong. Each participant received nightly eye drops but was randomly assigned to receive drops with 0.05 percent atropine, 0.01 percent atropine or a placebo. Families and clinicians didn’t know which treatment group the children were in.
A total of 353 children used their assigned eye drops for two years. Only about 25 percent of children who took 0.05 percent atropine eye drops, roughly 30 kids, developed myopia in at least one eye, compared with about 50 percent of those who used 0.01 percent atropine or placebo eye drops, around 60 kids in each group. The percentages in each group were similar for eye elongation not severe enough to be considered myopia.
“It’s a great first step to encourage us to explore more,” Saunders says.
Scientists will need to conduct studies in more diverse populations and environments to reach generalizable conclusions since the trial took place in only Hong Kong. Eye color may also influence dosing, as lighter-pigmented eyes might be more susceptible to side effects, including sensitivity to light.
How atropine slows myopia onset and progression remains a mystery. The medication might improve blood circulation in the eye, Yam says, but that’s just one existing hypothesis.
The new study was too short to suggest that atropine eye drops can prevent myopia. But an ongoing follow-up period in which participants continue taking the medication through their teenage years — when eye length stabilizes — will help the team understand if atropine eye drops can ward off the condition altogether.







