Fat Chance: Cancer drugs may also thwart obesity
A promising class of anticancer-drug candidates, which work by depriving growing tumors of needed blood vessels, also prevent obesity or cause dramatic weight loss in rodents. This discovery rests upon the unappreciated fact that fat tissue, like a tumor, requires an increased blood supply to grow, says Maria A. Rupnick of Brigham and Women’s Hospital in Boston, who led the study on the drugs.
“It’s clearly a potential way to think about treating obesity,” says Marc L. Reitman, director of obesity research at Merck Research Laboratories in Rahway, N.J. “I think the paper is very interesting, novel, and provocative.”
Rupnick works with M. Judah Folkman of Children’s Hospital in Boston, who originated the once-controversial idea that a growing tumor requires the creation of blood vessels in a process called angiogenesis. The new study stemmed from Rupnick’s desire to study angiogenesis in healthy adult tissues. The conventional view, however, was that because most adult tissues and organs maintain a stable size, adult animals rarely need new blood vessels except during specialized circumstances, such as wound healing and reproduction.
However, Rupnick had an epiphany several years ago while recalling her own decade-old work to isolate cells that form blood vessels in fat tissue.
“In that instant I realized, ‘Oh, my gosh, this is the best tissue we could possibly use to study blood vessel growth in a noncancerous system,'” she says. Fat “can grow and regress very substantially, very rapidly, and repeatedly, even in an adult.”
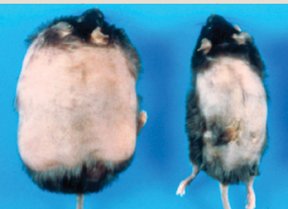
Rupnick subsequently confirmed that substantial blood vessel growth does occur in the fat tissue of mice that rapidly gain weight. And in the Aug. 6 Proceedings of the National Academy of Sciences, she, Folkman, and their colleagues report that some of the same angiogenesis inhibitors now being tested on cancer patients prevent excess weight gain in several different strains of obesity-prone mice.
The drugs did more than prevent new fat accumulation. They also triggered overweight mice to shed significant amounts of fat–up to half their body weight. “We were surprised by how much weight they lost,” says Rupnick.
She speculates that the blood vessels in existing fat tissue remain in a more malleable state than most other mature blood vessels. This would facilitate rapid expansion or shrinkage of the surrounding fat tissue. Tumors harbor similarly malleable blood vessels, which are vulnerable to angiogenesis inhibitors; most vessels elsewhere are unaffected. Consequently, in mice that lose weight, the drugs probably destroy preexisting blood vessels, as they do in tumors, and lead to the death of fat cells, Rupnick says.
Other data suggest the drugs also alter the metabolism of the mice so that they burn more fat than other energy sources.
Rupnick and her colleagues have ruled out the possibility that angiogenesis inhibitors simply made the mice sick or spoiled their taste for food. “We’ve treated animals for over a quarter of their lifespan, and they’re healthy and active,” she says.
One remaining puzzle centers on a mouse strain that typically develops life-threatening obesity as a result of genetically induced, insatiable hunger. As expected, such mice gained little weight when treated with angiogenesis inhibitors.
Surprisingly, however, the mutant mice also developed a normal appetite.
“I don’t know what’s causing the change in appetite, and I think we should have a better understanding of that,” says Rupnick. Answers to that question and others will be needed before angiogenesis inhibitors are tested against human obesity.
“Obesity drugs have to be incredibly safe,” says Reitman. “Personally, I think you have to know the mechanism by which an anti-obesity drug works in this current day and age.”
Folkman points out that the demand for angiogenesis inhibitors in cancer trials has already overwhelmed the limited supply.







