Few roads lead to meta, much to the chagrin of organic chemists trying to build compounds of particular shapes. Now researchers have found a simple way of getting molecules into the coveted meta position on an aromatic ring. The discovery, reported in the March 20 Science, opens the door to a flood of research that could yield products from new drugs to electronic materials, scientists say.
The find “combines new chemistry and recent developments with a very old problem,” says Robert Maleczka Jr. of Michigan State University in East Lansing, who wrote a commentary on the new work in the same issue of Science. “I would not be surprised to find another section in the textbooks.”
Chemist Matthew Gaunt of the University of Cambridge in England coauthored the work with graduate student Robert Phipps and says he hopes the find will help chemists more quickly make better molecules.
“We’re really happy about this,” Gaunt says. “The most exciting thing is where we can take it next.”
In 1825, English chemist Michael Faraday isolated benzene, and ever since then chemists have been piecing together how this ringed molecule’s properties govern its behavior. Benzene is what chemists call a simple aromatic ring — basically a bunch of atoms (carbon and hydrogen in benzene’s case) holding hands in a circle. The electrons are shared by everyone in the ring, existing in a cloud that spans the whole group.
Aromatic rings like benzene are known in the pharmaceutical world as “privileged structures.” They are flexible, reactive — great starter molecules. Forty of the top 50 prescription drugs in the country have aromatic rings, says Maleczka.
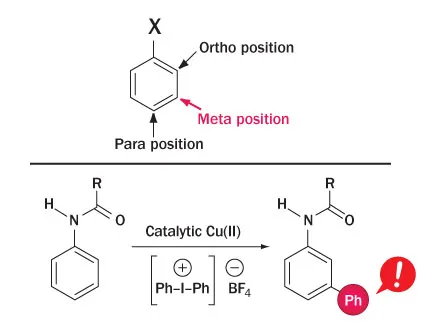
By the late 1800s, chemists had figured out that if they start with an aromatic ring like benzene that has a compound attached at the 12 o’clock position, additional compounds would always attach in one of three other places on the ring: ortho (2 o’clock), meta (4 o’clock) or para (6 o’clock). Over the years scientists have learned which molecules like to go to where — and what helper molecules can make that happen. Some metals, for example, are good at steering compounds to a particular sweet spot. These finds have allowed scientists to build drugs and all kinds of materials of particular, useful shapes.
But getting to meta has remained hard to do. As a result, investigations of some compounds may have been ignored in favor of ones that are easier to make, Maleczka says. “If you want to develop a new drug, chances are there is going to be a benzene ring in there somewhere.… It’s now very possible that we could expand the things we are making in a real practical way.”
The reactions that guide things to meta can be difficult and slow. Chemists have devised some tricks — but those techniques involve starting with clunky molecules that need to be broken down or using chemicals that have environmental baggage. Until recently, getting some molecules to meta required 10 steps and the explosive compound TNT as a starter material.
Gaunt and Phipps overcame meta-achieving difficulties by using copper and a form of iodine to get a compound known as a phenyl group to meta. It’s likely that the same procedure could be used with other rings and other groups, the researchers say.
The researchers started with a ring that, at 12 o’clock, had a group containing a nitrogen and a double-bonded oxygen hanging off the end. They mixed that in a flask with the group that they wanted to attach at the meta position, and added in copper 1 (CuI) and a form of iodine. With this set-up, the electrons in the bonded oxygen are attracted to the ortho position (2 o’clock). This attraction puts too many electrons into the ring’s cloud, so some of the electrons shift toward the meta position. Meanwhile the version of iodine gets copper all jazzed up, bumping it to copper 3 (CuIII). This version of copper wants electrons badly, so it moves to meta, bringing its phenyl group with it.
“Copper is kind of schizophrenic — it wants electrons, but once it starts getting some it also wants to do something to get rid of them,” says Maleczka.
So copper ditches the phenyl group to rid itself of two electrons. And tada! The phenyl group is attached at meta. Copper is free to react again, and everybody wins.
“A lot of what I like about the paper is the academic aspect,” Maleczka says. “We teach this to our sophomores all the time. It’s great finding something that breaks the rules.”






