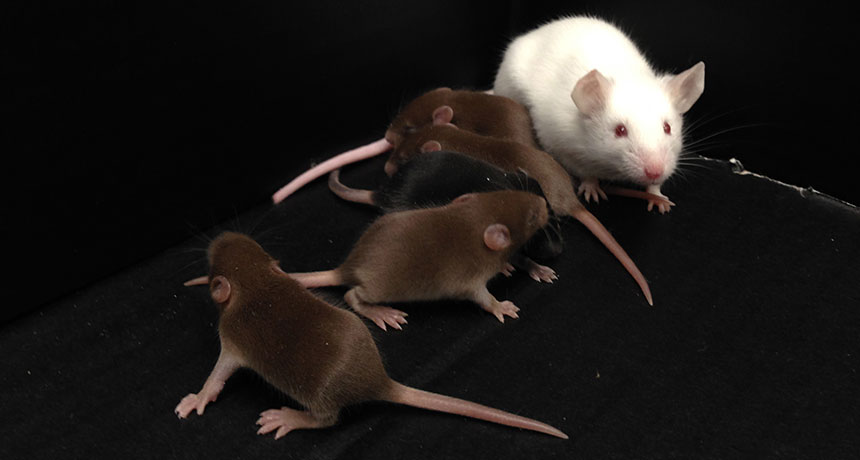
MADE MALE This male mouse (white) has no Y chromosome, but was able to father pups (brown) when scientists injected his immature sperm into eggs. Researchers made the mouse male by manipulating genes on other chromosomes.
Yasuhiro Yamauchi

MADE MALE This male mouse (white) has no Y chromosome, but was able to father pups (brown) when scientists injected his immature sperm into eggs. Researchers made the mouse male by manipulating genes on other chromosomes.
Yasuhiro Yamauchi