
GENE SCENE Zebrafish experiments that didn’t go as planned may help researchers find genes that can fill in for others in a pinch.
Novartis AG/Flickr(CC BY-NC-ND 2.0)
Zebrafish can find a way to compensate for a mutated gene, but artificial methods of inactivating the same gene are not so readily overcome, a new study suggests.
These findings, reported July 13 in Nature, add fuel to a technical debate among researchers about how to tell what a gene does in an organism.
An organism’s alternate strategy to compensate for a mutated gene can mask the gene’s absence. Zebrafish possess such a workaround for a mutation in a gene involved in blood vessel development, the new research finds. But fish with a normal copy of the gene can’t cope when researchers artificially block protein production from the same gene. Blocking protein production ought to have the same effect as a genetic mutation, but the fish handle the challenges differently, the researchers found.
If the same mechanisms that allow zebrafish to compensate for their defective genes are also working in humans, it could explain why some people with a genetic mutation develop severe disease while others with the same mutation are relatively healthy.
“It’s potentially really fascinating how genomes deal with their own weaknesses,” says Cecilia Moens, a developmental geneticist at the Fred Hutchinson Cancer Research Center in Seattle. She was not involved in the study.
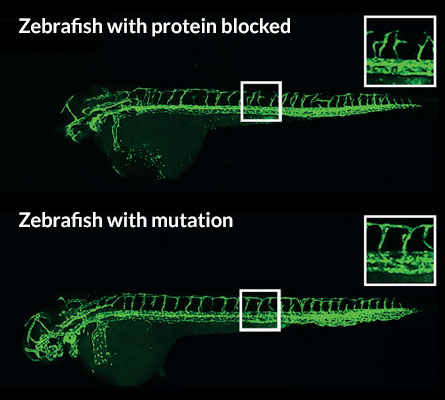
Many other researchers have experienced the same thing; DNA mutations have no effect or produce only mild changes in the fish, while stopping protein production with a morpholino created severe defects.
Morpholinos are synthetic molecules that zip together with RNAs from specific genes and gum up the works. That prevents RNA instructions from being used to build proteins or blocks other important interactions. RNA interference, which tampers with protein production by causing RNA to get diced into tiny bits, has also produced results contrary to those from mutations.
Many researchers have interpreted these discrepancies as side effects, or off-target effects, produced when morpholinos mess with RNAs other than the ones they are supposed to be inhibiting. The mutants create the “real” effect, say researchers including Moens and Nathan Lawson, a developmental biologist at the University of Massachusetts Medical School in Worcester.
But mutants may not be telling the whole story, Stainier says. Their tale can have hidden plots. Fish with mutations in egfl7 boosted activity of the Emilin2 and Emilin3 genes, the researchers found. The extra emilin proteins were able to fill in for the missing egfl7 protein and allow blood vessels to develop normally. The real effect of not having the egfl7 protein was unmasked when the researchers used the morpholino to stop its production, the researchers say.
Exactly how the fish compensate for the mutation is unknown. Stainier is also stumped about why the same mechanism doesn’t work when protein production is blocked.
Lawson says this may be a special case and that compensation is probably rare. “It’s going to take a few years for us to know how prevalent a mechanism this is.”
But developmental geneticist Stephen Ekker of the Mayo Clinic in Rochester, Minn., says such compensatory mechanisms are probably widespread, and may affect how human genetic diseases develop. “There are multiple states of normal,” says Ekker. “We can start measuring this molecularly.”







