The 2001 Nobel Prize in Chemistry, announced Oct. 10, honors research that led to
new chemicals, materials, and drugs, including widely used heart medicines and the
Parkinson’s disease treatment L-dopa.
Three chemists from different institutions share the $943,000 award. William S.
Knowles, retired from Monsanto in St. Louis, and Ryoji Noyori of Nagoya University
in Japan each win a quarter of the prize. K. Barry Sharpless of the Scripps
Research Institute in La Jolla, Calif., receives the other half.
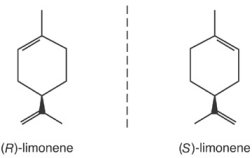
The award this year highlights research to control reactions that otherwise
produce mixtures of molecules in left- and right-handed forms, somewhat like left- and right-hand gloves. This property, which can greatly affect molecules’ traits,
is called chirality. The new honorees invented metal-based catalysts that promote
reactions that give products with just one handedness.
Knowles and Noyori are cited specifically for creating catalysts that can make a
chemical reaction, called hydrogenation, produce only one of two mirror-image
products. Sharpless is cited for related work with reactions called oxidations.
Before the Nobel winners’ work, chemists would try “with great difficulty” to
separate mixtures of chiral molecules, comments 1994 Nobel laureate George Olah of
the University of Southern California in Los Angeles. Inventing catalysts to make
only one version of a molecule in the first place is, in Olah’s words, “a very big
advance in science.”
Single-handed molecules are crucial in medicine, since proteins, amino acids, and
other biochemical targets of drugs exist naturally in only one of the two forms.
Ignoring chirality in biological settings can pose grave dangers. A mixture of
left- and right-handed forms of the antinausea drug thalidomide caused grievous
birth defects during the 1950s and 1960s. Although one form of the drug molecule
reduced morning sickness as intended, its chiral partner harmed many fetuses, the
Nobel committee notes.
The Nobel prize “came as pretty much of a surprise to me,” says Knowles, whose
research in the 1960s led to commercialization of the chiral amino acid L-dopa.
The Nobel committee woke him at 4 a.m. with news of the award, he says.
Sharpless tells Science News that he’s happy his 90-year-old father can see that
the science books purchased for his young son decades ago have paid off.
The 1980 synthesis of chiral molecules called epoxides–participants in many
reactions–was “a eureka moment,” Sharpless says. That work of his led to numerous
practical catalysts that are now commercially available.
“We all thought nature had a monopoly” on designing chiral molecules, he adds.
“But it turns out to be a whole lot easier than we thought.”
For more on the 2001 Nobel prizes, see the following articles:
“Nobel prizes mark 100th anniversary” (SN: 10/13/01, p. 230: Nobel prizes mark 100th anniversary)
“Nobel prize: Physiology or medicine” (SN: 10/13/01, p. 230: Nobel prize: Physiology or medicine)
“Nobel prize: Physics” (SN: 10/13/01, p. 230: Nobel prize: Physics)






