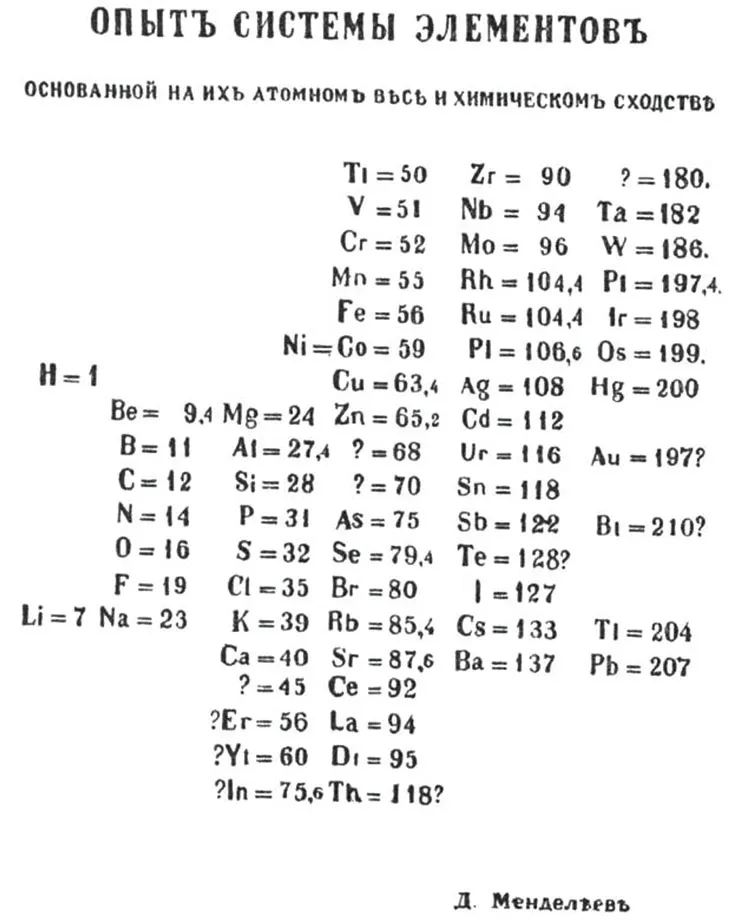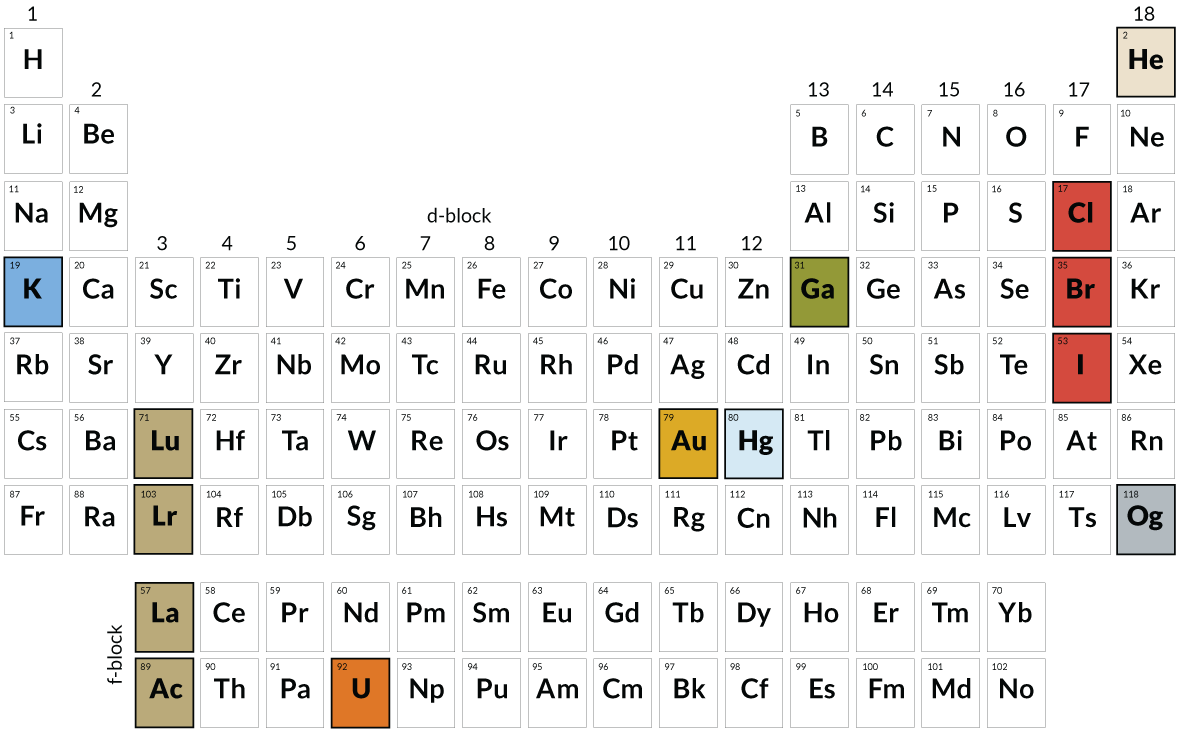How the periodic table went from a sketch to an enduring masterpiece
150 years ago, Mendeleev perceived the relationships of the chemical elements

REVOLUTIONARY Russian chemist Dmitrii Mendeleev (shown around 1880) was the first to publish a periodic table, which put the known elements into a logical order and left room for elements not yet discovered.
HERITAGE IMAGE PARTNERSHIP LTD/ALAMY STOCK PHOTO
Every field of science has its favorite anniversary.
For physics, it’s Newton’s Principia of 1687, the book that introduced the laws of motion and gravity. Biology celebrates Darwin’s On the Origin of Species (1859) along with his birthday (1809). Astronomy fans commemorate 1543, when Copernicus placed the sun at the center of the solar system.
And for chemistry, no cause for celebration surpasses the origin of the periodic table of the elements, created 150 years ago this March by the Russian chemist Dmitrii Ivanovich Mendeleev.
Mendeleev’s table has become as familiar to chemistry students as spreadsheets are to accountants. It summarizes an entire science in 100 or so squares containing symbols and numbers. It enumerates the elements that compose all earthly substances, arranged so as to reveal patterns in their properties, guiding the pursuit of chemical research both in theory and in practice.
“The periodic table,” wrote the chemist Peter Atkins, “is arguably the most important concept in chemistry.”
Mendeleev’s table looked like an ad hoc chart, but he intended the table to express a deep scientific truth he had uncovered: the periodic law. His law revealed profound familial relationships among the known chemical elements — they exhibited similar properties at regular intervals (or periods) when arranged in order of their atomic weights — and enabled Mendeleev to predict the existence of elements that had not yet been discovered.
“Before the promulgation of this law the chemical elements were mere fragmentary, incidental facts in Nature,” Mendeleev declared. “The law of periodicity first enabled us to perceive undiscovered elements at a distance which formerly was inaccessible to chemical vision.”
Mendeleev’s table did more than foretell the existence of new elements. It validated the then-controversial belief in the reality of atoms. It hinted at the existence of subatomic structure and anticipated the mathematical apparatus underlying the rules governing matter that eventually revealed itself in quantum theory. His table finished the transformation of chemical science from the medieval magical mysticism of alchemy to the realm of modern scientific rigor. The periodic table symbolizes not merely the constituents of matter, but the logical cogency and principled rationality of all science.
Laying the groundwork
Legend has it that Mendeleev conceived and created his table in a single day: February 17, 1869, on the Russian calendar (March 1 in most of the rest of the world). But that’s probably an exaggeration. Mendeleev had been thinking about grouping the elements for years, and other chemists had considered the notion of relationships among the elements several times in the preceding decades.
In fact, German chemist Johann Wolfgang Döbereiner noticed peculiarities in groupings of elements as early as 1817. In those days, chemists hadn’t yet fully grasped the nature of atoms, as described in the atomic theory proposed by English schoolteacher John Dalton in 1808. In his New System of Chemical Philosophy, Dalton explained chemical reactions by assuming that each elementary substance was made of a particular type of atom.
Chemical reactions, Dalton proposed, produced new substances when atoms were disconnected or joined. Any given element consisted entirely of one kind of atom, he reasoned, distinguished from other kinds by weight. Oxygen atoms weighed eight times as much as hydrogen atoms; carbon atoms were six times as heavy as hydrogen, Dalton believed. When elements combined to make new substances, the amounts that reacted could be calculated with knowledge of those atomic weights.
Dalton was wrong about some of the weights — oxygen is really 16 times the weight of hydrogen, and carbon is 12 times heavier than hydrogen. But his theory made the idea of atoms useful, inspiring a revolution in chemistry. Measuring atomic weights accurately became a prime preoccupation for chemists in the decades that followed.
When contemplating those weights, Döbereiner noted that certain sets of three elements (he called them triads) showed a peculiar relationship. Bromine, for example, had an atomic weight midway between the weights of chlorine and iodine, and all three elements exhibited similar chemical behavior. Lithium, sodium and potassium were also a triad.
Other chemists perceived links between atomic weights and chemical properties, but it was not until the 1860s that atomic weights had been well enough understood and measured for deeper insights to emerge. In England, the chemist John Newlands noticed that arranging the known elements in order of increasing atomic weight produced a recurrence of chemical properties every eighth element, a pattern he called the “law of octaves” in an 1865 paper. But Newlands’ pattern did not hold up very well after the first couple of octaves, leading a critic to suggest that he should try arranging the elements in alphabetical order instead. Clearly, the relationship of element properties and atomic weights was a bit more complicated, as Mendeleev soon realized.
Organizing the elements
Born in Tobolsk, in Siberia, in 1834 (his parents’ 17th child), Mendeleev lived a dispersed life, pursuing multiple interests and traveling a higgledy-piggledy path to prominence. During his higher education at a teaching institute in St. Petersburg, he nearly died from a serious illness. After graduation, he taught at middle schools (a requirement of his scholarship at the teaching institute), and while teaching math and science, he conducted research for his master’s degree.
He then worked as a tutor and lecturer (along with some popular science writing on the side) until earning a fellowship for an extended tour of research at Europe’s most prominent university chemistry laboratories.
When he returned to St. Petersburg, he had no job, so he wrote a masterful handbook on organic chemistry in hopes of winning a large cash prize. It was a long shot that paid off, with the lucrative Demidov Prize in 1862. He also found work as an editor, translator and consultant to various chemical industries. Eventually he returned to research, earning his Ph.D. in 1865 and then becoming a professor at the University of St. Petersburg.
Soon thereafter, Mendeleev found himself about to teach inorganic chemistry. In preparing to master that new (to him) field, he was unimpressed by the available textbooks. So he decided to write his own. Organizing the text required organizing the elements, so the question of how best to arrange them was on his mind.
By early 1869, Mendeleev had made enough progress to realize that some groups of similar elements showed a regular increase in atomic weights; other elements with roughly equal atomic weights shared common properties. It appeared that ordering the elements by their atomic weight was the key to categorizing them.
By Mendeleev’s own account, he structured his thinking by writing each of the 63 known elements’ properties on an individual note card. Then, by way of a sort of game of chemical solitaire, he found the pattern he was seeking. Arranging the cards in vertical columns from lower to higher atomic weights placed elements with similar properties in each horizontal row. Mendeleev’s periodic table was born. He sketched out his table on March 1, sent it to the printer and incorporated it into his soon-to-be-published textbook. He quickly prepared a paper to be presented to the Russian Chemical Society.
“Elements arranged according to the size of their atomic weights show clear periodic properties,” Mendeleev declared in his paper. “All the comparisons which I have made … lead me to conclude that the size of the atomic weight determines the nature of the elements.”
Meanwhile, the German chemist Lothar Meyer had also been working on organizing the elements. He prepared a table similar to Mendeleev’s, perhaps even before Mendeleev did. But Mendeleev published first.
More important than beating Meyer to the publication punch, though, was Mendeleev’s use of his table to make bold predictions about undiscovered elements. In preparing his table, Mendeleev had noticed that some note cards were missing. He had to leave blank spaces to get the known elements to properly align. Within his lifetime, three of those blanks were filled with the previously unknown elements gallium, scandium and germanium.
Not only had Mendeleev predicted the existence of these elements, but he had also correctly described their properties in detail. Gallium, for instance, discovered in 1875, had an atomic weight (as measured then) of 69.9 and a density six times that of water. Mendeleev had predicted an element (he called it eka-aluminum) with just that density and an atomic weight of 68. His predictions for eka-silicon closely matched germanium (discovered in 1886) in atomic weight (72 predicted, 72.3 observed) and density (5.5 versus 5.469). He also correctly predicted the density of germanium’s compounds with oxygen and chlorine.
Mendeleev’s table had become an oracle. It was as if end-of-game Scrabble tiles spelled out the secrets of the universe. While others had glimpsed the periodic law’s power, Mendeleev was the master at exploiting it.
Mendeleev’s successful predictions earned him legendary status as a maestro of chemical wizardry. But today, historians dispute whether the discovery of the predicted elements cemented the acceptance of his periodic law. The law’s approval may have been more due to its power to explain established chemical relationships. In any case, Mendeleev’s prognosticative accuracy certainly attracted attention to the merits of his table.
By the 1890s, chemists widely recognized his law as a landmark in chemical knowledge. In 1900, the future Nobel chemistry laureate William Ramsay called it “the greatest generalization which has as yet been made in chemistry.” And Mendeleev had done it without understanding in any deep way why it worked at all.
A mathematical map
In many instances in the history of science, grand predictions based on novel equations have turned out to be correct. Somehow math reveals some of nature’s secrets before experimenters find them. Antimatter is one example, the expansion of the universe another. In Mendeleev’s case, the predictions of new elements emerged without any creative mathematics. But in fact, Mendeleev had discovered a deep mathematical map of nature, for his table reflected the implications of quantum mechanics, the mathematical rules governing atomic architecture.
In his textbook, Mendeleev had noted that “internal differences of the matter that comprises the atoms” could be responsible for the elements’ periodically recurring properties. But he did not pursue that line of thought. In fact, over the years he waffled about how important atomic theory was for his table.
But others could read the table’s message. In 1888, German chemist Johannes Wislicenus declared that the periodicity of the elements’ properties when arranged by weight indicated that atoms are composed of regular arrangements of smaller particles. So in a sense, Mendeleev’s table did anticipate (and provide evidence for) the complex internal structure of atoms, at a time when nobody had any idea what an atom really looked like, or even whether it had any internal structure at all.
By the time of Mendeleev’s death in 1907, scientists knew that atoms had parts: electrons, which carried a negative electric charge, plus some positively charged component to make atoms electrically neutral. A key clue to how those parts were arranged came in 1911, when the physicist Ernest Rutherford, working at the University of Manchester in England, discovered the atomic nucleus. Shortly thereafter Henry Moseley, a physicist who had worked with Rutherford, demonstrated that the amount of positive charge in the nucleus (the number of protons it contained, or its “atomic number”) determined the correct order of the elements in the periodic table.
Atomic weight was closely related to Moseley’s atomic number — close enough that ordering elements by weight differs in only a few spots from ordering by number. Mendeleev had insisted that those weights were wrong and needed to be remeasured, and in some cases he was right. A few discrepancies remained, but Moseley’s atomic number set the table straight.
At about the same time, the Danish physicist Niels Bohr realized that quantum theory governed the arrangement of electrons surrounding the nucleus and that the outermost electrons determined an element’s chemical properties.

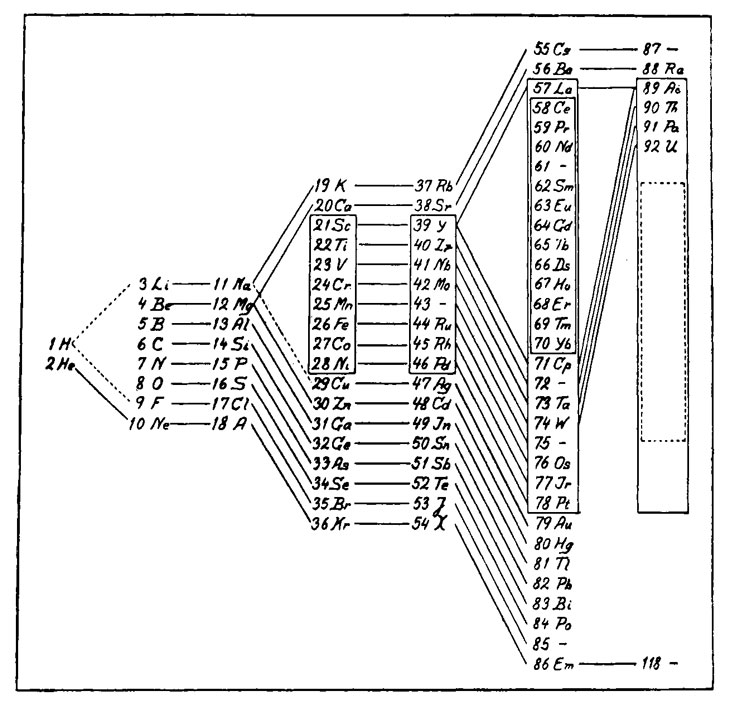
Similar arrangements of the outer electrons would recur periodically, explaining the patterns that Mendeleev’s table had originally revealed. Bohr created his own version of the table in 1922, based on experimental measurements of electron energies (along with some guidance from the periodic law).
Bohr’s table added elements discovered since 1869, but it was still, in essence, the periodic arrangement that Mendeleev had discovered. Without the slightest clue to quantum theory, Mendeleev had created a table reflecting the atomic architecture that quantum physics dictated.
Bohr’s new table was neither the first nor last variant on Mendeleev’s original design. Hundreds of versions of the periodic table have been devised and published. The modern form, a horizontal design in contrast with Mendeleev’s original vertical version, became widely popular only after World War II, largely due to the work of the American chemist Glenn Seaborg (a longtime member of the board of Science Service, the original publisher of Science News).
Seaborg and collaborators had synthetically produced several new elements with atomic numbers beyond uranium, the last naturally occurring element in the table. Seaborg saw that these elements, the transuranics (plus the three elements preceding uranium) demanded a new row in the table, something Mendeleev had not foreseen. Seaborg’s table added the row for those elements beneath a similar row for the rare earth elements, whose proper place had never been quite clear, either. “It took a lot of guts to buck Mendeleev,” Seaborg, who died in 1999, said in a 1997 interview.
Seaborg’s contributions to chemistry earned him the honor of his own namesake element, seaborgium, number 106. It’s one of a handful of elements named to honor a famous scientist, a list that includes, of course, element 101, discovered by Seaborg and colleagues in 1955 and named mendelevium — for the chemist who above all others deserved a place at the periodic table.