At Germany’s weeklong Hannover Fair last April, a camcorder monitored the crowds visiting the energy-exhibits section of the giant technology trade show. The device had no battery nor was it plugged into the wall. Instead, the palmsize camera got its power from a prototype fuel cell that transformed hydrogen gas and oxygen into water and electricity. The unusually compact fuel cell was “pretty reliable,” though not dependable enough to keep the camera running continuously, says Christopher Hebling of the Fraunhofer Institute for Solar Energy Systems in Freiburg, Germany, the alternative-energy laboratory that built the fuel cell.

In the same vast exhibit hall were examples of much larger fuel cells for cars, homes, and even factories (SN: 11/13/93, p. 314). Despite growing interest in such alternative energy sources, the introduction of fuel cells in those realms remains an uphill battle. Although clean and efficient, these systems, which have been under development for decades, remain expensive.
On the other hand, say technology analysts, if reliable fuel cells as small as the one in the camcorder–and smaller–could be mass-produced, consumers would snap them up. Users of cell phones, laptop computers, and other portable electronics are frustrated with having to recharge batteries every few hours, says Atakan Ozbek of Allied Business Intelligence in Oyster Bay, N.Y.
Fuel cells as small as a few centimeters across–known as micro fuel cells–could last 10 or more hours and be refueled in seconds, their promoters say.
For personal electronics, “people . . . desperately need the product,” says Robert K. Lifton of New York City–based fuel cell developer Medis Technologies.
Ozbek expects sales of the little power plants to reach 200 million units per year by 2008, at a price of $30 to $50 apiece.
That potential market is stimulating a broad range of efforts to develop micro fuel cells. Furthest along are so-called direct-methanol fuel cells, which have evolved from research funded by the Defense Advanced Research Projects Agency in the late 1980s and early 1990s. Today, several developers of these fuel cells are promising to roll out their first products–most likely, battery chargers–within the next year or so.
Nevertheless, direct-methanol fuel cells remain rife with problems ranging from getting flooded to drying out, and they produce uneven electrical output. “It’s a complicated system,” says Shimshon Gottesfeld of MTI MicroFuel Cells in Albany, N.Y.
Keenly aware of such shortcomings, many researchers are pursuing alternatives–some with surprising features. These include butane fuel cells with components that get scorching hot and a fuel cell that runs on an ingredient found in honey and beer.
Give it some juice
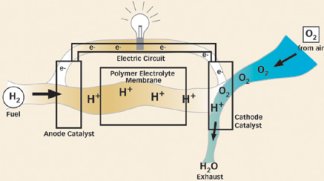
Today’s typical fuel cell, which can be as a big as a house, contains multiple electricity-generating units that are stacked like pancakes. Each of those units consists of two flat, metallic electrodes that sandwich a layer of electrolyte. The electrolyte layer, which can be liquid or solid, acts as a membrane allowing ions but not electrons to pass. This creates a voltage.
Although superficially similar to a battery, a fuel cell gets its energy in a different way. In a battery, chemical reactions produce current and consume the electrodes. In a fuel cell, however, chemical reactions consume fuel that comes from outside the cell, and the electrodes act only as catalysts for those reactions.
In general, fuel cells take in hydrogen and oxygen and convert them into water and electricity. However, sometimes the hydrogen is generated directly within the cell from another fuel, such as methanol.
At one electrode, the anode, molecules of hydrogen gas break down into electrons and positively charged hydrogen ions. Those ions–protons–then migrate through the electrolyte to the other electrode, the cathode.
Meanwhile back at the anode, the electrons from the hydrogen flow out of the fuel cell, where they serve as usable electric current that eventually returns to the cathode.
There, oxygen combines with the hydrogen ions and electrons to create water and heat.
Both electrodes incorporate precious-metal catalysts that spur the power-generating reactions.
The first fuel cell, made in 1839, used the liquid electrolyte sulfuric acid, and many modern fuel cells still have liquid electrolytes–usually held in a spongelike matrix. However, for some fuel cells, particularly the small ones, developers rely on solid, damp electrolytes. The most common one is known as a polymer-electrolyte membrane or proton-exchange membrane, both names conveniently boiling down to the acronym PEM.
Most current PEM fuel cells run on pure hydrogen, but the gas requires bulky storage canisters ill-suited to handheld appliances. To make a direct-methanol PEM cell, however, developers beefed up the catalysts in the electrodes to promote more-complex reactions, notably the combination of methanol and water to form carbon dioxide and the hydrogen needed to drive the cell.
Methanol, or methyl alcohol, is an inexpensive, hydrogen-rich liquid that could be sold in replaceable cartridges small enough for a cell phone. Potentially, a person could talk on his or her cell phone for 10 hours using one 10-to-20 milliliter cartridge.
Methanol is already used in windshield-washer fluid, and it’s the fuel for Indianapolis 500 race cars.
On the down side, methanol is toxic and flammable. Under current government rules, an electronic device containing it would be forbidden on a commercial aircraft. Besides that drawback, direct-methanol fuel cells face many technical hurdles.
A major headache is water management. Not only is water needed at the anode to react with methanol, but also the fuel cell makes water at the cathode. The more power a fuel cell makes, the faster it produces water and the warmer it gets. Since these fuel cells are not closed containers, that heat leads to the evaporation of some water from the cell. That’s only part of the water picture.
The fuel cell also needs moisture in the electrolyte as well as water at the anode to control the supply of methanol. Too high a concentration is undesirable because unused methanol migrates through the electrolyte to the cathode. When that happens, “you waste a lot of fuel–potentially more than half,” says Jesse Wainright of Case Western Reserve University in Cleveland. Fuel cell makers typically want about a 3 percent solution of methanol at the anode.
The outside temperature and humidity also influence the water distribution inside the fuel cell. If under humid conditions a cell has too much water at the cathode, oxygen can’t get to the electrode, and the fuel cell shuts down. In a dry climate, the heat from fuel cell operation can parch the electrode, also stopping the device.
“We’d love to be able to go from the Amazon jungle to the middle of Saudi Arabia and not have the performance and power output crash on us,” says Robert Hockaday of Manhattan Scientifics of Los Alamos, N.M.
To solve these and other problems of direct-methanol fuel cells, some research teams are outfitting their prototypes with dynamic controls, such as minuscule sensors, pumps, and sophisticated circuits. To make such fuel cells easy to mass-produce,
Motorola, for instance, has incorporated such components into a set of ceramic-based microchips that control fluid flows (SN: 8/15/98, p. 104: http://www.sciencenews.org/sn_arc98/8_15_98/bob1.htm), says Jerry Hallmark of the company’s research lab in Tempe, Ariz.
Other teams are striving to control their fuel cell’s operations by using solely passive processes, such as evaporation, diffusion, and capillary action. Though less efficient, these fuel cells would be simpler, cheaper to make, and less likely to break, Hockaday says.
Power play
Dozens of companies are developing direct-methanol and other types of fuel cells for handheld devices. The players range from small start-ups such as Polyfuel of Menlo Park, Calif., and Smart Fuel Cells of Brunnthal-Nord, Germany, to huge corporations including Samsung in Korea and Japan’s Toshiba and Casio. Because of intense competition, Gottesfeld says, “most information is kept close to the chest by all players.”
Although the direct-methanol fuel cell is the leader among micro–fuel cell designs, its drawbacks have left the field open to many potential challengers. Medis Technologies, where Lifton is chief executive officer, claims to have concocted an electrolyte that prevents alcohols from leaking to the cathode. The company’s fuel cell, which was developed by scientists from the former Soviet Union, will get power not from methanol, but ethanol laced with proprietary additives, says Lifton. “Our people have actually run the fuel cell on vodka,” he notes, adding that methanol works as well.
Richard I. Masel of the University of Illinois at Urbana-Champaign and his colleagues recently unveiled another way to prevent anode-to-cathode fuel crossover–by using formic acid for fuel. The chemical, which is secreted by ants and is also found in peaches, beer, and honey, transforms inside a fuel cell into a negatively charged compound that can scarcely pass through the electrolyte membrane. His team unveiled the device in August at a Detroit meeting for Defense Department–funded developers of tiny power supplies.
Another alternative fuel is sodium borohydride, a compound that can be made to release hydrogen gas far more efficiently than methanol does. It’s also nontoxic, notes Adam P. Briggs, a vice president of MillenniumCell in Eatontown, N.J. In a fuel cell, the compound turns into sodium borate, an ingredient of soap. Although this fuel is more expensive than methanol, running a cell phone for a month would still take less than a quarter’s worth of fuel, Briggs claims.
Other designs exploit the fact that a hot fuel cell generates more power than a cooler one because the chemical reactions go faster. On the other hand, hot cells demand cooling.
To turn up the thermostat on PEM cells, Wainright and Robert F. Savinell, who is also at Case Western Reserve, and their colleagues have made a novel, dry, solid-polymer electrolyte. They chemically modified polybenzimidazole, a plastic used to make fibers in protective clothing for firefighters and racecar drivers. The new membrane can tolerate temperatures above the boiling point of water. “The secret is that it doesn’t need water. It operates just fine perfectly dry,” Wainright says.
He and others, including Masel and Hallmark’s group, are also independently developing small, hot chambers external to a fuel cell that break down, or “reform,” hydrogen-rich fuels. Such so-called microreformers can extract hydrogen gas from methanol or other fuels and then feed it to the anode of a PEM cell.
Sossina Haile of the California Institute of Technology in Pasadena and her colleagues are developing one of the literally hottest prospects. Haile’s team specializes in making relatively cool-running versions (SN: 4/21/01, p. 247: Available to subscribers at Novel fuel cell gets hot, but not by a lot.) of scorching, solid-oxide fuel cells, which typically run at 800 to 1000C. The group’s new shot-glass-size unit consumes propane or butane fuel at about 500 to 600C.
Haile’s colleague Paul D. Ronney of the University of Southern California in Los Angeles is trying to make the fuel cell cool enough for consumer use by burying it deep in coiled heat-exchange tubes.
Other micro–fuel cell innovations also look promising. For example, Sony has created a dry electrolyte containing soccer-ball-shape carbon molecules called buckyballs. Another Japanese giant, NEC, reports manipulating carbon structures at the atomic scale to make better electrodes than those currently available. The team of Paul A. Kohl of the Georgia Institute of Technology in Atlanta is building methanol-vapor fuel cells small enough to sit on a microchip. The cells would both cool and power the chip.
To replace batteries, these many micro fuel cell technologies must not only clear technical hurdles but also demonstrate that they are inexpensive, safe, and dependable.
Manufacturing techniques are another prime concern, Hebling says. Fraunhofer researchers recently completed a yearlong study in which they found ways to automate nearly every step in the fabrication of their camcorder fuel cell.
On the other hand, many fuel cell developers say they have no intention of eliminating batteries. Rather, they expect to team their fuel cells with tiny batteries that will provide extra power as needed. Consumers probably won’t care whether future fuel cells are battery-assisted. What they’ll be watching is how many hours have passed since the last refill.





